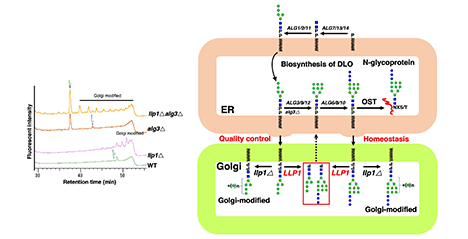
N-glycosylation, as the most important co-/post-translational modification of proteins, begins with the assembly of dolichol-linked oligosaccharide (DLO) on the ER membrane. Eukaryotic cells rarely accumulate DLO, however, immature and mature DLO structures are accumulated under some genetic and environmental conditions, thereby increasing a risk of abnormal N-glycosylation. Here, I describe that identify a novel gene encoding DLO pyrophosphatase responsible for the degradation of these DLO. ...and more
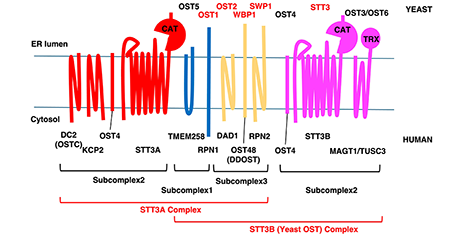
Asparagine (N)-linked glycans on proteins are one of the most common co- and post-translational modifications found not only in eukaryotes but also in bacteria and archaea. This reaction is carried out by enzymes called “oligosaccharyltransferases” (OSTs). While the active subunit often functions alone in bacteria and archaea, different subunit structures are known to exist in eukaryotic cells depending on the species. Recently, it has become clear that OSTs not only transfer glycans to proteins but also generate free glycans by hydrolysis of donor substrates, i.e., dolichol-linked oligosaccharides (DLOs). This hydrolysis reaction is significantly enhanced in mammals compared to budding yeast, suggesting that OSTs have evolved to more efficiently hydrolyze DLOs to form free N-glycans. Here, we provide an overview of what is known about OSTs and propose a hypothesis regarding the significance of their hydrolysis reaction. ...and more
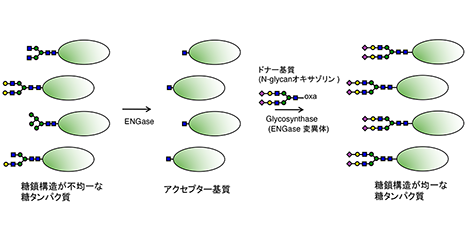
Glycans play important roles in protein function, stability, intermolecular interaction, and so on. These are highly related to glycan structures in many cases. However, the structure of the glycan attached to the specific glycosylation site usually varies in each molecule of glycoprotein, and this variation gives rise to the heterogeneity and functional differences between glycoproteins. Therefore, glycan remodeling aims to increase the homogeneity and strengthen the function and stability of the glycoprotein by attaching glycans of specific structure to the glycosylation sites of the glycoprotein. The glycan remodeling techniques have been widely developed, and one of them is the endo-β-N-acetylglucosaminidase (ENGase, EC 3.2.1.96)-based method. In connection with this, this review outlines the basic knowledge of glycan remodeling and the recently identified novel ENGases that recognize the non-reducing end of N-glycans and can hydrolyze multibranched complex type N-glycans. ...and more
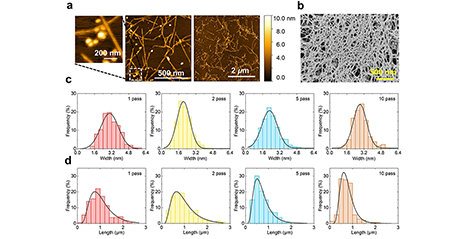
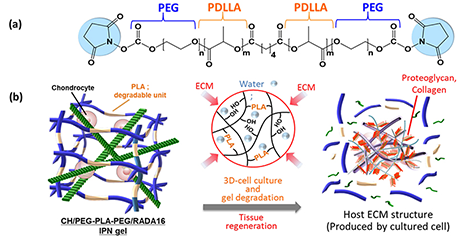
As a tissue engineering approach in regenerative medicine, we developed a novel hydrogel to serve as a scaffold for culturing cells in three dimensions and established a method for its simple synthesis. Here, hydrogel refers to a polymer chain network that contains water or other liquids. In vivo, cells proliferate and differentiate using the extracellular matrix (ECM), formed from various biomacromolecules such as collagen and hyaluronic acid, as a scaffold. Therefore, when culturing cells in vitro, a material serving as a scaffold is also required. Recently, a hydrogel called an interpenetrating polymer network (IPN), where multiple polymers intertwine to form independent, multiple network structures, has gained attention as a material mimicking the ECM. ...and more
AFood waste and food loss account for nearly 10% of global greenhouse gas emissions, seriously contributing to accelerating climate change. Coffee is one of these foods that create waste. Coffee is an agricultural product with high global consumption and large distribution volumes. Large quantities of spent coffee grounds (SCGs) are generated during coffee processing and consumption; SCGs are thus a common form of food waste. The majority of SCGs is disposed of in landfills, and approximately 11.14 million tons of SCG waste were generated between 2022 and 2023 according to statistics from the International Coffee Organization. SCGs are a promising biomass resource because they contain various components that can be used as raw material for fuels, biopolymers, and other products. Furthermore, SCGs are a model waste product for the “cross-economy,” a concept that redefines the use of resources and redesigns resource circulation among different industries and technological fields. In this paper, we outline the manufacturing process, structure, and physical properties of a novel nanocellulose derived from SCGs. ...and more