
Warren Knudson
Warren Knudson first became interested in connective tissue research during his graduate studies in the laboratory of Dr. Ed Conrad at the University of Illinois at Urbana Champaign. His Ph.D. work included the demonstration of chondroitin-4- and 6-sulfated disaccharide co-polymers that exist along the glycosaminoglycan chains of aggrecan. In 1981 he moved to Tufts University in Boston to begin postdoctoral studies in the laboratory of Dr. Bryan Toole. It was in Dr. Toole’s laboratory where his interest in hyaluronan began and, where he met his future wife, Dr. Cheryl B. Anderson. His work in this laboratory centered around the accumulation of hyaluronan associated with tumor tissues and receptors present on the tumor cell surface (CD44) that interact with hyaluronan. In 1985 he joined the connective tissue group in the Department of Biochemistry at Rush Medical College in Chicago, being recruited by the department chairman, Professor Klaus Kuettner. His work continued along the lines of tumor connective tissue until connections were made between the tumor cell hyaluronan receptors and those present on articular chondrocytes. Currently, as an Associate Professor of Biochemistry, the focus of his laboratory is centered around the role of chondrocyte CD44 in the internalization and degradation of hyaluronan.

Cheryl B. Knudson
Cheryl Knudson received her B.A. degree from Pomona College in Claremont, California and her Ph.D. from the University of Southern California. Her Ph.D. work with the late Dr. Stephen Meier showed the segmental emigration of cranial neural crest cells and the involvement of mesodermal pattern in their distribution. She also studied the role of hyaluronan in neural crest cell migration, and this directed her interest to postdoctoral studies in the laboratory of Dr. Bryan P. Toole at Tufts University in Boston. With Professor Toole she investigated the role of hyaluronan-cell interactions during limb development, describing the hyaluronan receptor activity expressed during chondrogenesis. She joined the faculty in the Department of Biochemistry at Rush Medical College in Chicago in 1985 and is currently an Associate Professor of Biochemistry. This department has developed around a central research theme of connective tissue, under the leadership of Professor Klaus E. Kuettner. Although she continues her research into cell-matrix interactions during development, her interests include hyaluronan-cell interactions in cartilage homeostasis, aging and disease.
Hyaluronan serves a variety of functions within the extracellular matrix of nearly all tissues with some of these functions including direct effects on cell behavior.a How cells organize their extracellular matrix and how that matrix feeds back to affect cell metabolism involve specific cell-matrix interactions—interactions that are mediated by cell surface matrix receptors. Several cell membrane-localized receptors (a.k.a., “hyaluronan binding proteins”) have been identified including; CD44, RHAMM b, IVd4 a and the Liver Endothelial Cell clearance receptor (LEC receptor). This list is likely to grow in the future, as is indicated by preliminary studies from many laboratories. Nevertheless, probably the most common hyaluronan receptor and the most studied to date is CD44. CD44 gets its current name from its identity with a family, or cluster of common leukocyte antigens (Clusters of Differentiation, CD) as defined by the International Workshop on Human Leucocyte Differentiation Antigens. However, prior to its designation as CD44, other names had been given to this same protein, including Pgp-1 (phagocytic glycoprotein-1), In[Lu]-related p80, Hermes antigen, HUTCH-1, gp90, gp85, H-CAM, and ECMRIII (extracellular matrix receptor type III), as well as the common functional name, “HA receptor.” The latter name alludes to the fact that the activity and properties of CD44 as a plasma membrane-associated hyaluronan binding protein were known long before its structure, name, and sequence identity were determined.
a See review by Toole in this series.
b See review by Turley in this series.
In many early studies on cell-cell aggregation, hyaluronan was found to mediate cross-bridging between cells. Such cross-bridging suggested the existence of membrane-localized hyaluronan binding sites. Small fragments of hyaluronan occupying individual hyaluronan binding sites or representing hyaluronan too small to bridge sites between cells, inhibited cell aggregation. Excess high-molecular-mass hyaluronan also inhibited this aggregation because of receptor saturation. Hyaluronan mediated homotypic as well as heterotypic cell-cell aggregation, provided both cell types expressed hyaluronan receptor activity. For example, the homotypic aggregation of normal lymphocytes, as well as the aggregation of lymphoma cells with macrophages or B-cells with bone marrow stromal cells, were induced by small concentrations of hyaluronan.
In the late 1970s, a membrane protein of approximately 85 kDa on SV3T3 cells was identified to be most likely responsible for mediating the binding of hyaluronan to the cell surface and cross-bridging (aggregation) of adjacent cells.1 This membrane protein/receptor on intact cells displayed a high apparent binding affinity for hyaluronan (a Kd of 10-9 M). This strong binding affinity was due to multivalent interactions with multiple receptors, as the binding affinity of individual receptors after membrane-solubilization was significantly lower. The same cells displayed saturable binding of hyaluronan (~1.3x106 Dal) with a Bmax of 5-10 mg hyaluronan bound/106 cells. The binding activity also had other, more unique properties. It was calcium-independent, stable to mild fixation with agents such as formaldehyde or glutaraldehyde, and exhibited increased binding affinity with increased ionic strength.1 Binding of hyaluronan to this 85-kDa protein was effectively inhibited by small hyaluronan oligosaccharides with a minimum size of a hexasaccharide. On intact cells, other glycosaminoglycans such as chondroitin sulfate or heparan sulfate showed little capacity to compete with hyaluronan for binding. However, once detergent solubilized, the hyaluronan receptor lost its binding specificity, and chondroitin sulfate could readily compete with hyaluronan binding. Interestingly, chondroitin, the non-sulfated form of chondroitin sulfate, can function as an effective, albeit still weak, competitor of hyaluronan binding on intact cells.2
All of these properties were characterized initially using rodent cell lines such as SV3T3 and endothelial cells. However, later studies showed that cells from a variety of tissues and species display nearly identical hyaluronan receptor characteristics. These include embryonic chick limb bud chondrocytes, adult bovine and human articular chondrocytes, and hamster alveolar macrophages, as well as several highly invasive human bladder carcinoma cells. Nonetheless, the hyaluronan receptor resisted attempts at its purification to homogeneity. Finally, Underhill et al.,3 identified an antibody (K-3) that can block hyaluronan-mediated cell-cell aggregation as well as the binding of radiolabeled hyaluronan to endothelial cells. It was of no surprise that the K-3 antibody reacted with an 85-kDa endothelial cell membrane protein. However, subsequent experiments proved highly fortuitous when the same antibody reacted with a well-characterized, sequenced, 85-kDa leukocyte homing receptor antigen named CD44. Thus, in one experiment the identity of this elusive hyaluronan receptor became known. From this point on, as they say, “everything is history.”
In reality, at about this same time it was already becoming apparent from other investigators that CD44 was a likely hyaluronan receptor. Sequence analysis predicted that the CD44 homing receptor is a membrane-intercalated glycoprotein with a hydrophobic transmembrane domain and an extracellular domain with ~40% homology to link protein, a hyaluronan binding protein in the extracellular matrix of cartilage. In the March 24, 1989, issue of Cell, two separate groups of investigators reported the sequence homology between link protein, the proteoglycan aggrecan, and the N-terminal extracellular portion of the lymphocyte homing receptor. Thus this exciting correlation brought together investigators interested in the biological activity of hyaluronan-cell binding and those studying CD44 as a lymphocyte homing receptor, all of which led to multiple reports that CD44 is the principal cell surface receptor for hyaluronan.
Subsequent studies confirmed the identity of CD44 as the protein responsible for hyaluronan binding. In many cell systems, hyaluronan binding was blocked using anti-CD44 specific monoclonal antibodies, and cells such as lymphocytes were shown to bind to hyaluronan-coated surfaces. In our studies, when COS-7 cells were transfected with constructs that contained human pCD44, the cells gained the capacity to bind hyaluronan and to assemble a pericellular matrix or “coat” in the presence of exogenously added hyaluronan and aggrecan. Further, these activities were stable to mild glutaraldehyde fixation, and the matrix assembly was inhibited by hyaluronan hexasaccharides. All cells, from chondrocytes to tumor cells, that we have tested that are able to assemble coats using exogenously added hyaluronan and aggrecan, also express abundant levels of CD44. Thus, although CD44 may not be responsible for all observed hyaluronan binding activity, there is also little doubt that it represents a major contributor to the binding of hyaluronan to the surface of cells.
A. Protein Domain Structure
CD44 is a single-pass transmembrane glycoprotein consisting of four functional domains, (Fig 1). The distal extracellular domain is the region primarily responsible for the binding of hyaluronan. The membrane-proximal extracellular domain is the primary site of alternative splicing of CD44 mRNA that produces the many isoforms of CD44. The CD44 transmembrane domain is fairly typical of most single-pass membrane glycoproteins. However, site-directed mutatagenesis studies as well as work with detergent-solubilized hyaluronan receptors suggest that associated lipids or accessory membrane proteins, interacting with this domain, modulate hyaluronan binding as well as CD44 interaction with the cytoskeleton. In most isoforms of CD44, a 70-amino-acid cytoplasmic domain or “tail” is also expressed. This cytoplasmic domain exhibits protein motifs that indicate a capacity for interaction with cytoskeletal proteins as well as the potential for intracellular signaling.
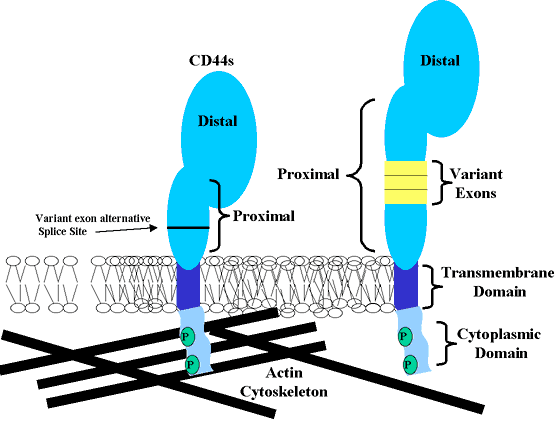
Fig. 1 Protein Domains of CD44.
The four principal protein domains of CD44 are diagrammed including (1) the distal extracellular domain (link protein-homologous domain), (2) the membrane proximal extracellular domain, (3) the transmembrane domain, and (4) the intracellular cytoplasmic domain. Also shown is an isoform of CD44 containing protein extensions within the membrane proximal extracellular domain (3 exons shown in yellow). These protein extensions are absent in CD44s, the most common isoform of CD44.
B. CD44 Gene / mRNA
The CD44 gene consists of 20 exons (19 exons in earlier literature, exons 6a and 6b have been reclassified as exons 6 and 7, to make 20 exons total), (Fig 2). Although a single gene located on the short arm of human chromosome 11 encodes CD44, multiple mRNA transcripts that arise from the alternative splicing of 12 of the 20 exons have been identified. The standard and most prevalent form of CD44 (termed CD44s) consists of a protein encoded by exons 1-5,16-18, and 20 (the exons shown in dark blue in Fig. 2). Since this form is also the predominant form on hematopoetic cells, CD44s is also known as CD44H. CD44s exhibits the extracellular domains (exons 1-5 and 16), the highly conserved transmembrane domain (exon 18), and the cytoplasmic domain (exon 20) (Fig. 1). The 1482 bp of open reading frame mRNA for human CD44s results in translation of a polypeptide chain of ~37 kDa. Post-translational addition of N-linked and O-linked oligosaccharides contribute to the ~85-kDa molecular mass of the final CD44 protein as estimated by SDS-PAGE.
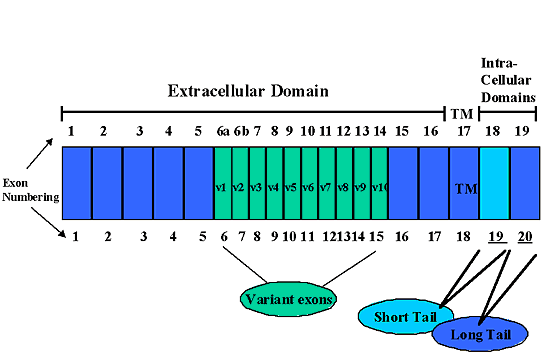
Fig. 2 Alternative Splicing of CD44 Exons.
The exons that encompass the CD44 gene are numbered and diagrammed. The top line of numbering illustrates a numbering system that uses 19 exons for CD44. The bottom line is based on the current numbering system, which uses 20 exons for CD44. The exons used for CD44s, the most common isoform of CD44, are shown in dark blue. Turquoise-colored exons denote the variant exons that exhibit extensive rearrangements due to alternative splicing, resulting in a myriad of variant isoforms of CD44. Alternative splicing may also occur within exons encoding the intracellular domain of CD44. Either exon 19 (light blue) or exon 20 (dark blue) are utilized, resulting in a short cytoplasmic tail isoform of CD44 or the complete cytoplasmic domain isoform, respectively.
Several isoforms of CD44, ranging in size from 80 to 250 kDa, arise by the alternative splicing of so-called “variant” exons into the extracellular domain of CD44 (see Figs. 1 and 2). Alternative splicing of exons 6-15, also named variant exons v1-v10, increases the length of the exposed receptor, possibly modulating some of its functions. For example, transfectants expressing CD44v6+7 gained the capacity to bind chondroitin sulfate as well as hyaluronan. The nomenclature of these variant isoforms is not yet standardized. Thus in specific areas of study the variant isoforms have user-friendly names such as epican, descriptive names such as gp116 (CD44v10 found in endothelial cells), and variant number designation names, CD44vn, with the “n” corresponding to the variant exons inserted, e.g., CD44v7-9. Some of the variant isoforms of CD44, such as CD44v3 (epican) contain Ser/Gly sites with chondroitin sulfate or heparan sulfate glycosaminoglycan chains, converting CD44 to a proteoglycan (Fig. 3). Although other potential Ser/Gly sites for glycosaminoglycan chains are present within CD44H and CD44v10, only the site within exon 8 (variant exon 3) has been found to contain chains.
Another site of alternative splicing of CD44 is within the intracellular domain. Either exon 19 or 20 are differentially expressed due to alternative splicing and represent two variations of the intracellular “tail” portion of the molecule (Fig. 2). In actuality, the 5’ end of exon 19 differs from exon 20 by only one base pair. However, this difference results in a chain termination stop codon and the expression of a truncated cytoplasmic domain with only four amino acids (Fig. 3). Thus the alternatively spliced message containing exon 19 in lieu of exon 20 generates a “short-tailed” form of the CD44 protein. The lack of intracellular signaling motifs and of protein domains necessary for interaction with cytoskeletal components has fueled intense speculation over the role of this CD44 isoform. Although the expression of exon 19 in most cells is relatively rare, we have recently identified CD44exon19 mRNA transcripts in human chondrocytes—with the same cells also expressing transcripts for CD44s (CD44exon20).
Little information is available concerning the regulation of CD44 gene expression. CD44 is transcriptionally upregulated by proinflammatory cytokines such as IL-1 as well as by growth factors such as EGF, TGF-beta and osteogenic protein-1 (BMP-7). IL-1 increases CD44 mRNA and protein expression levels in both chondrocytes and vascular smooth muscle cells. In chondrocytes, the increased CD44 expression results in an increased capacity for hyaluronan binding, internalization, and degradation of hyaluronan. In smooth muscle cells, IL-1 induced elevation of CD44 increased the capacity of these cells to bind hyaluronan. This increased expression of CD44, together with increases in hyaluronan that are elaborated in atherosclerotic lesions, may contribute to the development of a pathological lesion.c EGF upregulates CD44 expression in fibroblasts, several tumor cell types, and epithelial cells undergoing stratification. Therefore, it is interesting that a putative 120-bp EFG response element/transcription factor binding site was identified in the CD44 gene of fibroblasts, 450 bp upstream of the RNA initiation site.4 It is likely that this response element will be found in other cell types as well.
c See review by Wight in this series.
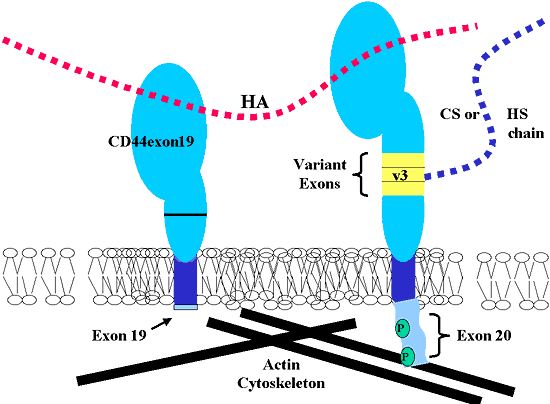
Fig. 3 Examples of Two Alternatively Spliced Isoforms of CD44.
To the left is a representation of exon 19 containing CD44 (CD44exon19). Introduction of a termination codon within exon 19 results in a truncated cytoplasmic domain with only four amino acids extending into the cytoplasm. To the right is a representation of a CD44-containing protein extension within the membrane-proximal domain due to the alternative splicing of three variant exons, including exon 8, also known as variant exon 3 (v3). The expression of the v3 domain extension allows for the addition of a heparan sulfate or chondroitin sulfate side chain.
C. Post-translational Modifications
As with other transmembrane glycoprotein receptors, CD44 undergoes extensive post-translational modifications including phosphorylation, glycosylation, and addition of glycosaminoglycan chains in some CD44 isoforms. All of these modifications are being actively explored as potential modulators of hyaluronan binding or of other CD44 functions. The word “modulate” is used because few of these modifications appear to “mediate or regulate” CD44 activity in an absolute sense. For example, some investigators maintain that CD44 glycosylation is a necessary prerequisite for hyaluronan binding, while others find that recombinant nonglycosylated CD44 still binds hyaluronan. Phosphorylation/dephosphorylation of serine/threonine residues of the cytoplasmic tail of CD44 represent dynamic modifications that suggest a role for “inside-out” regulation of CD44 function as well as “outside-in” signal transduction. Phosphorylation has also been shown to influence linking and unlinking the tethering of CD44 to the actin cytoskeleton. In fact, in several cell types, CD44 exists in two pools: one that is extracted using mild detergent conditions and the other, presumably CD44 tightly associated with the cytoskeleton, that requires more drastic conditions for solubilization. Phosphorylation may control the switch between these two pools of CD44, which in turn may regulate the capacity of CD44 to bind hyaluronan.
D. Molecular Interactions Required for Hyaluronan Binding
Several attempts have been made to define the exact site and molecular interactions involved in hyaluronan binding to CD44. Early on, as described above, it was known that CD44 had a high degree of homology to the B-loop structure of link protein and aggrecan. However, unlike link protein and aggrecan, CD44 exhibits only one half of the tandem repeat structure. Also, the fact that the binding of hyaluronan to CD44 can usually be competed effectively with hyaluronan hexasaccharides suggests that the active binding site of CD44 is smaller than that of the link protein or aggrecan which bind hyaluronan decassacharides. Peach et al.5 made a careful analysis of two clustered regions of basic amino acids, one within the distal portion of the region of homology to the link protein and another closer to the proximal domain (Fig. 4). Two major findings came from this work. First, as expected, basic amino acids within the distal, link protein-homologous domain, appear to be essential for CD44 binding to hyaluronan. One residue in particular, arginine 41, was considered critical for binding. However, their data also demonstrated that the other, more proximal cluster of basic amino acids was also necessary (Fig. 4). Mutations in either domain significantly reduced CD44 binding to hyaluronan. CD44-immunoglobulin fusion proteins, truncated to contain only the distal link protein-homologous domain (i.e., no proximal domain), displayed a low capacity to bind to hyaluronan coated plates. In addition, point mutation of any of the four clustered basic arginine residues also reduced hyaluronan-binding activity. The investigators suggested that the two domain regions may work cooperatively, in some fashion, to bind hyaluronan. Their studies also suggested that the anti-CD44 monoclonal antibody IM7 recognized an epitope near the proximal domain site necessary for hyaluronan binding. This accords well with our observations that IM7 bound to chondrocytes can block CD44-hyaluronan interactions (without CD44 shedding) even though IM7 binds outside of the link protein homologous domain.
In more recent studies, a three-dimensional model of CD44 has been generated based on homology of CD44 to the protein TSG-6 (TNFalpha-stimulated glycoprotein-6). TSG-6, like CD44, is another member of the link protein-homologous superfamily. However, in the case of TSG-6, the three-dimensional solution structure of the link protein domain of the protein has been determined and was found to be similar to a calcium-dependent lectin fold such as that found in a rat mannose-binding protein. Using the site of this putative fold domain for direction and Arg-41 as a starting point, Bajorath et al.6 generated and tested additional conservative and nonconservative amino acids point mutations. The studies revealed a large surface region of the link protein-homologous domain of CD44 likely to be responsible for hyaluronan binding. As shown in Fig. 4, a particular cluster of amino acids (Tyr-42, Arg-78, and Tyr-79), together with the previously described Arg-41, form the likely critical region for hyaluronan binding. Mutations in this region inhibit CD44’s capacity to bind to hyaluronan, and inhibit the binding of anti-CD44 monoclonal antibodies that block hyaluronan binding. In the predicted model structure, all four of these amino acids run along a ridge on the surface of the CD44 protein. This same study revealed that only Tyr-42 is conserved between CD44 and TSG-6. It was thus concluded that, despite the similarities, details of the protein-carbohydrate interactions within the link protein superfamily may significantly differ. This finding may help explain differences in binding affinities, specificities, and competitor oligosaccharide size of hyaluronan binding to CD44 versus its binding to TSG-6, link protein, or aggrecan.d
d See review by Day in this series, forthcoming in 1999.
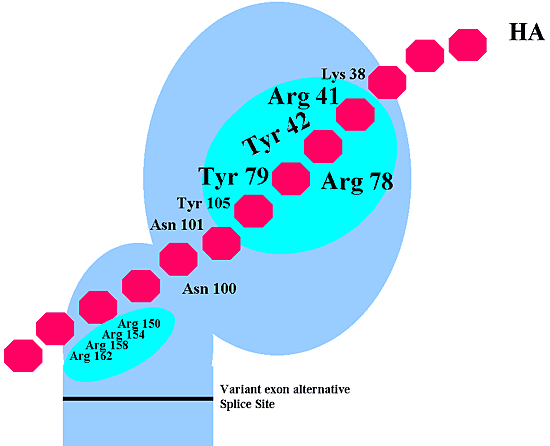
Fig. 4 The Putative Hyaluronan Binding Domains of CD44.
The two putative hyaluronan binding domains within the distal extracellular (link protein-homologous) domain, as well as the membrane-proximal extracellular domain (distal to the variant exon alternative splice site) are shown in light purple. The amino acids that are believed to be directly responsible for hyaluronan binding are shown. Within the major hyaluronan site of the distal link protein-homologous domain, the four critical amino acids (Arg-41, Tyr-42, Arg-78, and Tyr-79) are highlighted in large type. We extend special thanks to Dr. A. Day for his review of this figure.
CD44 is known to participate in a wide variety of cellular functions, including cell-cell aggregation, retention of pericellular matrix, matrix-cell and cell-matrix signaling, receptor-mediated internalization/degradation of hyaluronan, and cell migration. However, even though this list is long, it only includes functions that involve CD44-hyaluronan interactions. For example it does not include the role of CD44 as a lymphocyte antigen or as a cell surface proteoglycan. Although many of these functions are well characterized, how cells regulate their usage of CD44 remains a mystery. Regulation of function may involve some of the aspects described above, namely, use of particular CD44 isoforms, post-translational modifications of CD44, or differential association of CD44 with other proteins such as cytoskeleton-linkage intermediates. Examples of a few of these functions will be discussed in the following sections.
A. CD44 Anchors Hyaluronan / Proteoglycan-rich Pericellullar Matrices
CD44-hyaluronan interactions mediate cell aggregation via cell-matrix cross-bridging. However, when the hyaluronan levels reach the point of saturation of available CD44 receptor sites, the cells disaggregate.a The now independent cells remain apart, cocooned within their own cell-associated or “pericellular” matrix. These cell-associated pericellular matrices, often extending to a distance equal to the cell diameter, can be visualized around living cells in vitro by the use of a particle exclusion assay. Chondrocytes are one of the best examples of such cells (Fig. 5). These cells synthesize sufficient levels of hyaluronan and aggrecan so as to maintain a constitutive cell-associated matrix. This native cell-associated matrix can be displaced by the addition of hyaluronan hexasaccharides or by treatment with a dilute solution of Streptomyces hyaluronidase (Fig 5A and 5B insets). If these reagents are removed from the chondrocytes, the cells will resynthesize and reassemble a native pericellular matrix within 4-12 hours. However, reassembly can be prevented by incubation of the recovering cells in the presence of anti-CD44 antibodies such as IM7, chondroitin, hyaluronidase, or hyaluronan hexasaccharides (but not chondroitin sulfate hexasaccharides). Results such as these have led to the hypothesis that the hyaluronan/proteoglycan-rich cell-associated matrix is tethered to the plasma membrane of chondrocytes (and many other cell types) via the interaction of hyaluronan with CD44.
The retention of hyaluronan to the plasma membrane does not occur exclusively by interactions with receptors. It is clear that on a variety of cell types, including chondrocytes, hyaluronan is also bound to the plasma membrane through interaction with the hyaluronan synthase.a For example, prior to chondrogenesis, chick limb bud mesenchymal cells display prominent pericellular matrices that are Streptomyces hyaluronidase sensitive but not displaceable with hyaluronan oligosaccharides. However after chondrogenesis, the newly differentiated chondrocytes continue to exhibit a prominent matrix, but one that is now displaceable by the oligosaccharides. In the latter cells, greater than 70% of the cell-associated 35S-labeled aggrecan is also released into the culture medium in the presence of hyaluronan oligosaccharides. Thus, inhibiting CD44/hyaluronan interactions through the use of anti-CD44 antibodies or with hyaluronan hexasaccharides provides one method that can be used to distinguish between the two mechanisms for cell surface anchoring of hyaluronan. Another approach to define CD44-mediated anchorage is: (1) to remove all endogenous matrix via Streptomyces hyaluronidase treatment, (2) to fix the “matrix-free” cells with glutaraldehyde to inhibit all endogenous synthesis of hyaluronan and proteoglycan, and (3) to do binding assays on the fixed cells using exogenously added hyaluronan. We have shown that chondrocytes treated in such a fashion bind saturable levels of radiolabeled hyaluronan and, assemble a cell-associated matrix in the presence of added hyaluronan plus aggrecan, similar to their native matrix even when visualized by electron microscopy.2 This matrix assembly can be blocked by certain anti-CD44 antibodies, hyaluronan oligosaccharides, chondroitin or, treatment with Streptomyces hyaluronidase. In subsequent studies, we determined that any cell type expressing comparable levels of CD44, including tumor cells, capillary endothelial cells, or pCD44-transfected COS-7 cells, can assemble a similar pericellular matrix using exogenously added hyaluronan and proteoglycan. As with the chondrocytes, these matrices will assemble on live or fixed cells and can be displaced by agents such as hyaluronan hexasaccharides.
Most of the studies concerning hyaluronan-dependent matrix retention by anchorage to CD44 have been done with cell cultures. This always leaves open the question as to whether such interactions occur in vivo. This is difficult to address because most methods used to liberate cells from tissues, by definition, destroy the modes of interaction of cells with their extracellular matrix. As one approach to address this question, embryonic chick tibia were treated with a purified collagenase in the presence of 20% fetal bovine serum, a source of inhibitors of other potential proteases. Individual and small clusters of chondrocytes were isolated with a large proportion of their cell-associated matrix intact and tightly associated with their plasma membranes. However, following treatment of the liberated cells with hyaluronan hexasaccharides, these native, cell-associated matrices were competitively displaced and lost from the cells. (see C. Knudson in 7). The similarities between properties of these freshly isolated cells and cultured cells strongly suggest that CD44-hyaluronan interactions occur in vivo and are likely to be responsible for retention of hyaluronan-enriched matrices.
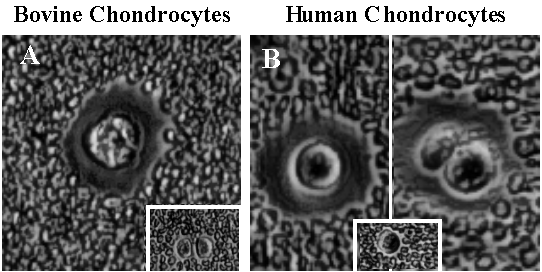
Fig. 5 The CD44-Anchored Cell-Associated Matrix of Chondrocytes.
Bovine and human normal articular chondrocytes were grown as primary cultures in alginate beads for 5 days following isolation from cartilage and then released as single cells by treatment with sodium citrate. The citrate causes the depolymerization of the Ca++ dependent alginate matrix. The cells were allowed to attach as monolayer cultures overnight. The culture medium was then removed and replaced with a suspension of particles, fixed horse red blood cells. After allowing 15 minutes for the particles to settle, the cultures were viewed by phase contrast microscopy using Varel optics. As shown in panels 5A and 5B, both bovine and human articular chondrocytes exhibit an extensive cell-associated pericellular matrix. Clustered cells share a cell-associated matrix as seen in the second group of human chondrocytes shown in the right half of panel 5B. Insets in panels 5A and 5B depict bovine and human chondrocytes, respectively, treated for 2 hours with hyaluronan hexasaccharides prior to visualization using the particle exclusion assay. We extend special thanks to Dr. Yoshihiro Nishida of our laboratories for these micrographs.
B. CD44 Mediates Communication Between the Cell and Its Matrix
The fact that CD44 is a transmembrane receptor with an extensive cytoplasmic domain automatically suggests that CD44 can communicate cell-matrix interactions into the cell (outside-in signaling) and can alter matrix in response to intracellular signals (inside-out signaling). The cytoplasmic domain of CD44 can undergo changes in phosphorylation, changes in association with the underlying cytoskeleton, and potentially, changes in interaction with adjacent CD44 molecules. However, results in this area and models developed from them are confusing and often contradictory. In one study, the deletion of all 70 of the amino acids of the cytoplasmic domain completely abolished hyaluronan binding. (see Lesley in 7) However, in a different study, lymphoma cells expressing CD44 with a truncated cytoplasmic domain continued to bind hyaluronan.9 Another study detailed how the ankyrin-binding motif in the cytoplasmic domain of CD44 was required for hyaluronan binding.10 On the other hand, mutants with deletions in the ankyrin-binding domain showed no change in hyaluronan binding capacity.9 Perhaps some alterations communicate changes in the functional usage of CD44 rather than having a direct effect on the capacity of CD44 to bind hyaluronan.
Although only speculation, one clue to understanding CD44 communication may be to consider the function of multiple CD44 interactions as opposed to individual receptor events. With traditional receptors such as the insulin receptor, binding of a single molecule of ligand to a single receptor induces conformational changes that transduce a signal into the cells. This may or may not be the case for CD44. The ligand, of course, is hyaluronan. The binding of hyaluronan, with multiple binding sites along its length, to CD44 may induce the clustering of receptors, or at least, generate a structural ordering of these receptors within the membrane. Because of its linkage to the underlying cytoskeleton, extracellular ordering of CD44 could result in a matching intracellular organization. Continuing downstream, changes in the organization of the cytoskeleton are known to cause the release or sequestration of a variety of secondary signaling intermediates (e.g., catenins, FAK, Src, p56, p185, Rho, PKC, IP3), that lead to subsequent changes in cellular metabolism. The process could also work in reverse, i.e., disruption of the cytoskeletal organization disperses CD44 in the membrane, which in turn modifies the capacity of CD44 to bind or otherwise organize extracellular hyaluronan. Thus, although more laborious a mechanism as compared to hormone-receptor models, this does provide a suitable means of cell-matrix communication. Is there precedence for such a model?
Several reports have observed three functional categories of CD44: nonbinding CD44, nonbinding CD44 unless activated, and constitutively active CD44 with respect to hyaluronan binding. What differentiates these three categories of CD44 is not entirely clear. Most cells of connective tissue, as well as some proliferative epithelial cells, appear to express constitutively active CD44. Many lymphocytes express nonbinding CD44 that can be activated by the use of bivalent anti-CD44 antibodies. The conclusion drawn in this case is that some form of clustering or ordering is a required prerequisite for CD44 to bind hyaluronan. Unfortunately, the identity of the natural activators of CD44 in such cells remains unknown. Nonetheless, these results suggest that an ordered networking of CD44 is likely to be required for its function as an hyaluronan receptor.
The fact that many connective tissue cells exhibit a constitutively active form of CD44 implies that either different forms of CD44 are being expressed or that CD44 is already ordered within the cell membrane as it is expressed. Given that CD44s is the predominate isoform expressed by most cell types, the latter suggestion may be more likely. In a study of bovine chondrocytes by immuno-electron microscopy, we found a regular repetitive pattern of anti-CD44 staining at the cell surface with a periodicity of 41-45 nm.2 Interestingly, anti-aggrecan staining that localized close to the cell surface also exhibited a repetitive pattern with a slightly longer periodicity of 60-70 nm. These results suggest that CD44 involved in binding the extracellular hyaluronan/aggrecan-rich matrix occurs, not as clustered receptors per se, but as a highly ordered array at the cell surface.
What of the intracellular side of this model? Does CD44 have an association with the underlying cytoskeleton and is this association dynamic enough to allow for cell-matrix communication? Following the addition of hyaluronan to T lymphoma cells, there is a rapid appearance of CD44 clusters and the co-localization of cytoplasmic ankyrin, an actin-binding protein, beneath these patches. The addition of hyaluronan to T lymphoma cells also produces an increase in intracellular calcium levels which may activate kinases (e.g., the PKC family of kinases) resulting in the phosphorylation of serine/threonine residues present on the intracellular domain of CD44 (Fig. 6). Phosphorylation of CD44 is known to increase the binding of ankyrin to the cytoplasmic domain of CD44.10 In addition, phosphorylation of CD44 appears to increase its capacity to bind hyaluronan. Conversely, phosphorylation-defective mutants of CD44 expressed in T lymphoma cells display reduced hyaluronan binding.11 Whether the effect of phosphorylation is due to direct conformational effects on CD44 structure or to an indirect cytoskeleton-stabilizing effect is unclear at present. However, in another study, phosphorylation incompetent mutants of CD44 did not exhibit altered hyaluronan binding or receptor distribution into detergent-soluble and cytoskeletal-associated (detergent-insoluble) pools.8 In the reverse, disruption of the cytoskeleton by reagents such as cytochalasin reduces hyaluronan binding in endothelial cells and a loss of hyaluronan-rich matrix in chondrocytes. Thus, although some reports contradict such a model, it is clear that at least in some cell types, binding of hyaluronan induces some form of ordering, which, in turn, results in reconfiguations of the underlying cytoskeleton. The likely mediator of this process, namely, differential phosphorylation of CD44, can potentially provide the dynamic change necessary for cell-matrix communication. For example, macrophages appear to exhibit two pools of CD44 with respect to phosphorylation. In resting peritoneal macrophages, the two pools are balanced between the phosphorylated and non-phosphorylated forms. However, in elicited macrophages, there is a shift such that greater than 90% of the CD44 becomes phosphorylated. In chondrocytes, we have found that removal of hyaluronan and the hyaluronan/proteoglycan-rich matrix via Streptomyces hyaluronidase treatment results in a reduction in phosphorylated CD44, a reduction that is reversed by subsequent addition of exogenous hyaluronan.
Studies by our laboratories and by others have documented the utilization of two families of actin-binding proteins responsible for linking the cytoplasmic domain of CD44 to the cortical actin cytoskeleton (Fig. 6). One family is called “ERM” to designate its component proteins (ezrin, radixin, moesin and merlin). The other is called the “ankyrin” family. As discussed above, upregulation of the kinase activity responsible for CD44s Ser/Thr phosphorylation enhanced CD44 association with ankyrin in T lymphocytes. However, the 140-kDa CD44 expressed in endothelial cells (CD44v9+v1) interacts with the cytoskeleton via ERM proteins while CD44v10 expressed on endothelial cells interacts with ankyrin for linkage to the cytoskeleton. We have found that chondrocytes express a component of the ERM family, namely, moesin, as well as ankyrin. However after the chondrocytes are lysed and solubilized, only ankryin co-immunoprecipitates with CD44s. Whether both of these linkage systems operate together or selectively (possibly for different functions) is not known at present but remains an exciting possibility.
Clustering of CD44, or the lack thereof, may also help explain the differential response of some cells to hyaluronan of different sizes. Recent work with macrophages showed that fragmented hyaluronan, but not the high-molecular-mass form, signals the production of IL-1, TNF-alpha, IGF-1, and iNOS by the macrophages. (see Noble in 7 ). Production of peroxynitrite by the reaction of NO and superoxide can degrade hyaluronan, generating fragments that would perpetuate the pathological cycle of inflammation. Other investigators have demonstrated that soluble hyaluronan oligosaccharides, of a defined size range, stimulate not only a migratory response of endothelial cells but, directed migration (e.g. chemotaxis). Low concentrations of hyaluronan oligosaccharides (6-20 monosaccharides) stimulate endothelial proliferation, are angiogenic in chorioallantoic membrane assays, and stimulate chemotactic migration. (see West in 7). Soluble native hyaluronan and hyaluronan oligosaccharides of larger sizes have no chemotactic influence on these cells. Whether such hyaluronan oligosaccharides exist in tissues is only speculation, but the disparate influence of hyaluronan of different molecular weights supports the hypothesis that the monovalent interaction of hyaluronan oligosaccharides may affect different signals from those resulting from the multivalent clustering interactions of hyaluronan with multiple receptors.
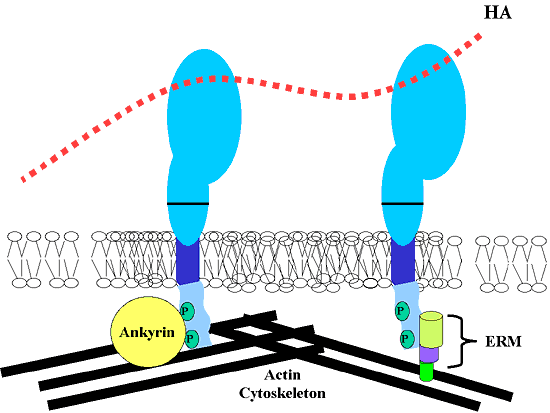
Fig. 6 The Actin Binding Proteins Associated with CD44.
CD44 is known to link to the actin cytoskeleton through its interaction with particular actin-binding proteins, binding proteins associated with either the ankyrin family (depicted in the left model of CD44) or the ERM family (depicted in the right model of CD44).
C. CD44 Mediates the Internalization of Hyaluronan, Leading to its Degradation
The turnover and removal of hyaluronan from the extracellular matrix occurs via local catabolism and/or drainage into the lymphatic system for catabolism in regional lymph nodes. It is estimated that only ~10% of the hyaluronan within the effluent lymph exits the nodes to enter into the circulation, where it is cleared by cells of liver sinusoids expressing hyaluronan-binding LEC receptors. Although the amount of hyaluronan turnover in some tissues such as epidermis is high, there is little evidence to support the presence of an extracellular, neutral pH active, hyaluronidase activity. Thus, except for possible free-radical mechanisms of extracellular hyaluronan depolymerization, local turnover of hyaluronan must occur intracellularly within lysosomes via a low pH active hyaluronidase. In addition, numerous studies also suggest that hyaluronan internalization is mediated via matrix receptors—including CD44 (Fig 7A).
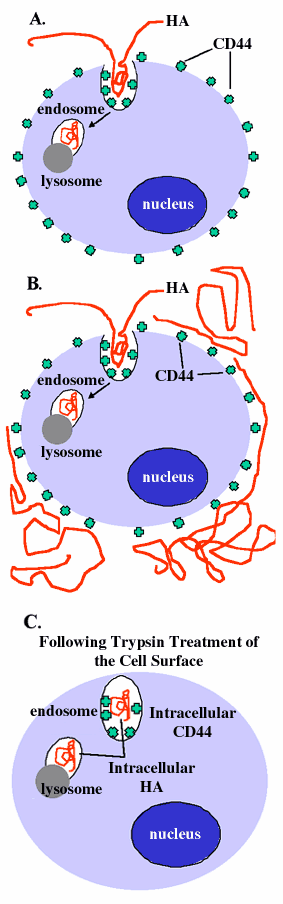
Fig. 7 CD44-Mediated Internalization and Degradation of Hyaluronan.
A portion of the hyaluronan, bound at the cell surface via interaction with CD44, is likely internalized by invagination of the plasma membrane, as shown in panel 7A. The nature of this internalization step has not, as yet, been fully characterized but is known not to represent the classical clatherin-coated vesicle. This invagination would become an endosome and subsequently fuse with a lysosome, completing the degradation of the hyaluronan. When exogenous fluorescein-hyaluronan is incubated with cells expressing unoccupied CD44 receptors, detected label will reflect extracellular as well as intracellular, internalized hyaluronan (panel 7B). Following removal of extracellular hyaluronan by treatment with Streptomyces hyaluronidase or removal of the entire matrix plus membrane proteins by trypsin treatment, intracellular hyaluronan is revealed (panel 7C). Actual cells as diagrammed in panel 7B are shown in Fig. 8A, 8C, and 8E; cells diagrammed in 7C are shown in Fig. 8B, 8D, and 8F.
Two cell types that exhibit prominent binding of hyaluronan via CD44, alveolar macrophages and SV-3T3 cells, internalize surface-bound hyaluronan, leading to its complete degradation. (see Fraser in 7). An example of such internalization is shown in Fig. 8A and 8B. Following 12-24 hours of incubation with fluorescein-conjugated hyaluronan, intense staining of SV3T3 cells can be seen (Fig. 8A). In order to delineate intracellularly localized hyaluronan, the cells were treated with trypsin or Streptomyces hyaluronidase (Fig. 8B and 8C). As can be seen in Fig. 8B, most of the labeled probe is removed, and what remains are small clusters of trypsin-resistant hyaluronan, apparently sequestered within intracellular vesicles. We have found that chondrocytes also can bind and internalize hyaluronan via CD44. Fig. 8C and 8D depict adult bovine articular chondrocytes incubated with exogenous fluorescein-hyaluronan before (Fig. 8C) and after treatment with trypsin (Fig. 8D). As with the SV3T3 cells, the intracellular hyaluronan accumulated in clusters or patches indicative of intracellular vesicles. Fluorescein-labeled dextran, a polysaccharide of similar molecular weight, did not bind or become internalized under similar conditions indicating that internalization of hyaluronan in all of these studies does not occur via simple fluid phase pinocytosis. Further, the internalization of hyaluronan by the chondrocytes was inhibited by excess unlabeled hyaluronan, hyaluronan hexasaccharides, or incubation with anti-CD44 antibodies. Thus, hyaluronan added to cells in solution can bind to the cell surface via interaction with CD44, and a portion subsequently undergoing endocytosis.
In order to provide better characterization, a 3H-labeled hyaluronan was used for the internalization studies. Autoradiography of chondrocytes incubated with 3H-labeled hyaluronan, before and after either trypsin or hyaluronidase-treatment, showed nearly identical patterns as those shown in Fig. 8C and 8D, e.g., following trypsinization, grains were localized within small clusters within the cells.12 From the summation of cpm, we estimate that ~5% of the labeled hyaluronan probe bound on the extracellular cell surface was internalized within 24 hours of incubation. Analyses of the intracellular pool displayed two size classes of label, one that eluted in the void volume of a Sepharose CL-2B column (i.e., >1 x 106 Da), and one that eluted in the total volume of the column (i.e. degradation products). The generation of these small, extensively degraded products was inhibited by the presence of the lysomotropic agent chloroquine. Therefore, the intracellular degradation of hyaluronan occurs within a low pH environment, such as that of the lysosome.
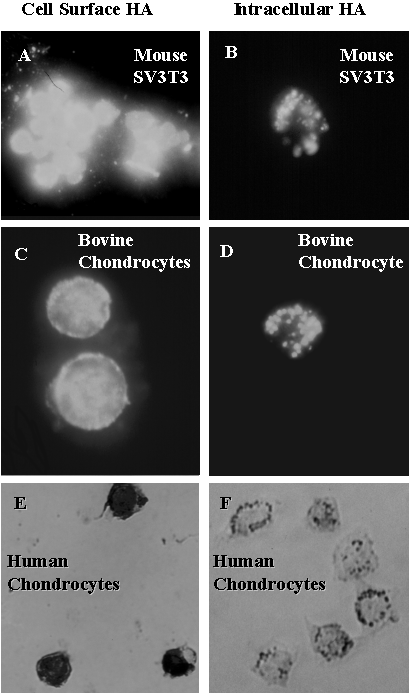
Fig. 8 CD44-Mediated Internalization of Hyaluronan in SV3T3 Cells and Chondrocytes.
Bovine and human normal articular chondrocytes were grown as primary cultures in alginate beads for 5 days following isolation from cartilage and then released as single cells by treatment with sodium citrate. The bovine chondrocytes were pre-treated with Streptomyces hyaluronidase to remove endogenous matrix and, together with the SV3T3 cells (grown directly in monolayers), the cells were incubated for 24 hours with fluorescein-conjugated hyaluronan (see Ref. 12 for details). Following incubation with the labeled ligand, one aliquot of the cells was washed and observed directly by fluorescence microscopy. The other aliquot was treated with 0.25% trypsin for 40 minutes to remove all extracellularly exposed matrix and surface protein, washed, and similarly observed by fluorescence microscopy. The human chondrocytes following release from alginate beads, were either: (1) fixed, permeabilized and directly stained with a biotinylated HABP and developed (panel 8E) or (2) treated with 0.25% trypsin for 40 minutes to remove all extracellularly exposed matrix/surface protein, washed, and similarly fixed, stained with the biotinylated HABP and developed (panel 8F). Panel 8F was overexposed digitally to demonstrate intracellular grains and panel 8E was overexposed by the same level. We extend special thanks to Ying Dang and Dr. Yoshihiro Nishida from our laboratories for SV3T3, bovine and human chondrocyte work, respectively.
It is clear, then, at least a portion of the hyaluronan bound to CD44 extracellularly is internalized and delivered to the lysosome for degradation. But does CD44 mediate the entire hyaluronan internalization event? In other words, is CD44 also internalized, and if so, does the hyaluronan remain bound to CD44 following internalization? Preliminary evidence from our laboratory suggests that CD44 is internalized and that CD44 and hyaluronan can be co-localized within intracellular vacuoles. Bovine articular chondrocytes were incubated with either phycoerythrin-anti-CD44 antibody or fluorescein-labeled hyaluronan. Following 3 hours of incubation to allow time for endocytosis, the chondrocytes were trypsinized to remove all cell surface-bound material. Small, punctate accumulations of fluorescence of both types co-localized within the cytoplasm as observed by conventional fluorescence and confocal microscopy, indicating internalization of the both tagged ligands. In another study, the Z-scan utility of the confocal microscope was used to validate the intracellular location of the antibody-tagged CD44 which was labeled extracellularly. The internalized cell surface-tagged CD44 represented ~20% of the total tagged CD44 before trypsin treatment.
In all of the above studies, exogenous labeled hyaluronan has been added to Streptomyces hyaluronidase-treated cells. This treatment is used to remove all endogenous hyaluronan bound to CD44 (as well as hyaluronan/PG-rich matrix in the case of chondrocytes) and expose the maximal number of hyaluronan binding sites. Is there also turnover of endogenous CD44-bound hyaluronan within a native cell-associated matrix? This question was addressed by the use of a specific morphological probe for hyaluronan, namely, a biotinylated hyaluronan (HA) binding protein (biotin-HABP) complex, namely the G1 domain of aggrecan and the link protein. In this study, adult normal human articular cartilage chondrocytes were cultured in alginate beads for 5 days and then released from the beads by treatment with sodium citrate as a calcium-chelating agent. As shown in Fig. 8E, extracellular hyaluronan accumulated in the cell-associated matrix stained prominently with the biotin-HABP probe. After, the cell-associated matrix was removed by Streptomyces hyaluronidase treatment, identical cells were stained for hyaluronan. At the identical exposure level that was optimal for panel 8E, only a hint in intracellular staining was noticed. While a longer exposure than that for untreated cells was required to enhance the streptavidin reaction product, intracellular vesicles were seen clearly positive for hyaluronan (Fig. 8F).
Interestingly, keratinocytes also bind and internalize hyaluronan, utilizing a CD44-dependent mechanism.e Using a biotinylated HABP probe and confocal microscopy, this group has also demonstrated intracellular accumulation of hyaluronan in these cells. In all of these systems, more data will be required to determine whether CD44-mediated internalization of hyaluronan is the major pathway for hyaluronan turnover or one that exists more as a “salvage” pathway. However, one of the notable phenotypes observed following the selective antisense transgene suppression of CD44 in mouse keratinocytes was the abnormal accumulation of hyaluronan in the superficial dermis as well as in the corneal stroma.13 Thus, in many tissues, CD44-mediated internalization of hyaluronan may be the primary mechanism for the turnover and catabolism of hyaluronan.
e See review by R. and M. Tammi in this series.
D. Function of CD44 / Hyaluronan Interactions in Tumor Invasion and Metastasis
In the last several years there has been intense interest in the association of CD44 expression and tumor progression/metastasis. It is not surprising that many malignant cell types overexpress CD44 because CD44 is a receptor expressed by many of the relevant cells prior to their transformation. However, during differential screening for epitopes present on a metastatic rat pancreatic carcinoma cell line versus a non-metastatic cell line, Gunthert et al., isolated a novel alternatively spliced isoform of CD44 termed CD44v6. (see W. Knudson, in ref 7). This variant CD44 contained an inserted amino acid sequence within the extracellular domain of the molecule. This sequence is derived from the addition of exon 11 (Fig. 2) due to alternative splicing. The expression of exons 6-15 is relatively rare, with v6 seldom expressed in normal cell types (e.g., v6 expression occurs transiently during B-cell activation). When an antibody specific to the variant CD44 isoform (CD44v6) was co-injected with the metastasizing cells, metastatic growth of the pancreatic carcinoma was inhibited, and host survival was prolonged. Interest in this CD44 variant peaked when it was found that transfection of nonmetastatic tumor cells with CD44v6 enhanced the cell’s efficiency for metastasis to regional lymph nodes. Since these initial observations, numerous studies have documented the prevalence as well as diagnostic/prognostic value of CD44 variant isoforms in human cancers, including the expression of alternatively spliced combinations of the v3, v6, and v9 isoforms of CD44. As expected there are also reports to the contrary. For example, the targeted disruption of CD44 in murine lymphosarcoma cells had no effect on their invasiveness or metastatic potential. 14
A necessary next question, especially in relation to this review, is whether binding to hyaluronan is a necessary component of CD44’s positive function in invasion and/or metastasis. An importance of CD44-hyaluronan binding would seem reasonable, given that several tumors, particularly those of human solid cancers, are often enriched in hyaluronan. However, two opposite views are suggested for the role of elevated tumor-associated hyaluronan: (1) the neo-synthesized hyaluronan-rich matrix provides a highly hydrated migration-promoting environment or (2) the hyaluronan-rich environment secures a viscous inhibitory barrier to cell migration. If the former is correct, enhanced expression of CD44H or CD44 variants could facilitate haptotactic-like locomotion through the tumor-associated hyaluronan-rich matrix. The tumor cells may also utilize RHAMM expressionb in the presence of hyaluronan, to assist in cell locomotion. If hyaluronan serves as a viscous barrier to migration, CD44 may also facilitate migration. A few investigators have correlated the cellular capacity for CD44-mediated endocytosis and degradation of hyaluronan as assessed in vitro to tumor metastatic aggressiveness. That is, the malignant cells that are best equipped to internalize and degrade hyaluronan appear to be the most efficient at metastasis. The interpretation is that the tumor-associated hyaluronan barrier is breached effectively by cells with a capacity to bind, internalize, and degrade this glycosaminoglycan. The two functions of CD44 may also work in combination namely, the binding and endocytosis of a substratum ligand as part of the mechanism for cell locomotion. The relationship between endocytosis capacity and its involvement in tumor progression remain to be defined better. This may require methods to inhibit CD44-mediated endocytosis selectively and to determine whether tumor cells maintain hyaluronan-mediated locomotion both in vivo and in vitro.
To address these questions more directly, investigators have established model systems for cell migration. Thomas et al. demonstrated that the stable transfection of human melanoma cells with pCD44 resulted in an enhanced motility of the cells on hyaluronan-coated surfaces as compared to the parental, CD44-negative, cells.15 This enhanced motility was inhibited either by anti-CD44 antibodies or by the presence of a CD44 ligand competitor, in this case a soluble CD44-immunoglobulin fusion protein. Thus, hyaluronan can function as support for the locomotion of tumor cells mediated via CD44. In another recent study, Yu et al., attempted to answer this question by transfecting a CD44-positive murine mammary carcinoma cell line (TA3/St) with cDNA encoding soluble CD44.a The transfectants spontaneously release soluble CD44, which in turn, competes with the activity of endogenous cell surface-localized CD44. After tail vein injection into syngeneic mice, control cells formed massive lung metastases. However, metastases were reduced to near zero when the transfectants expressing soluble CD44 were examined. With control cells or transfectants, when lung metastases became established, stimulated deposition of hyaluronan was observed. These data support the suggestion that, in vivo, some form of tumor cell CD44-mediated interaction is important for the stable establishment and growth of distant metastases. Whether the critical partner in this interaction with CD44 is hyaluronan, although highly likely, still remains to be determined definitively.
The above discussion should leave the reader with the notion that CD44 is an important, key molecule, especially as it relates to cell matrix interactions. It is thus somewhat startling that CD44-null mice are viable and do not demonstrate any obvious morphological defects.16 It is possible that other proteins may compensate for the function of CD44 in the knock-out mice. One recently reported candidate hyaluronan receptor is LYVE-1.17 However, the selective antisense transgene suppression of CD44 in keratinocytes, achieved using a tissue-specific promoter strategy, resulted in an obvious skin phenotype13 not observed in the knock-out mouse. The skin of the keratinocyte-selective mice was highly hydrated and easily torn. Closer examination revealed an aberrant accumulation of hyaluronan in the epidermis, likely due to inhibited CD44 receptor-mediated endocytosis of hyaluronan necessary for turnover. The suppression of CD44 expression also inhibited keratinocyte proliferation. Clearly, more research is needed to validate the significance and multiple roles of CD44 and/or the identity of other compensatory receptors that are expressed in its place. If other receptors come into play, it will be necessary to determine whether or how they function coordinately with CD44 under physiological conditions.
As has been discussed in previous monographs in this series, many cells are confronted with an extracellular matrix rich in hyaluronan. For many years the deposition of extracellular matrix, especially matrices rich in hyaluronan was viewed as setting a stage into which the cells could act their parts. The current view has changed. Now cells and matrix interact together, modulating each other and functioning as a unit. The key to this new view has been the discovery of cell surface matrix receptors, providing the means for interaction and communication between matrix and cells. For many receptors, such as the integrins, one receptor is sometimes used to interact with multiple ligands. In the case of CD44, it is likely that there is one ligand, hyaluronan, but numerous functions related to hyaluronan. Understanding CD44 will serve to direct future paradigms of matrix receptors and their role in cell-matrix interactions.