
Tony Day is a structural biologist who has worked on hyaluronan-protein interactions for the last seven years. He obtained a B.A. in Chemistry from Oxford University in 1985 followed by a D.Phil. in Biochemistry in 1988. His doctoral research focused on the structural characterization of factor H, a control protein of the human complement system. From 1991 to 1998 he worked as an Arthritis Research Campaign Research Fellow in the laboratory of Professor Iain D. Campbell (Department of Biochemistry, University of Oxford), and it was during this time that he developed his interest in hyaluronan-binding proteins. This work led to the determination of the first three-dimensional structure of a Link module, a domain found in many hyaladherins. Dr. Day is now a Senior Scientist at the Medical Research Council's Immunochemistry Unit (Oxford) and is engaged in research aimed at understanding the molecular basis of protein-hyaluronan interactions and the role of TSG-6 (an inflammation-associated hyaladherin) in extracellular matrix remodeling and leukocyte adhesion.
Hyaluronan has diverse biological roles in vertebrates. For instance, it acts as a vital structural component of connective tissues,a forms loose hydrated matrices that allow cells to divide and migrate (for example during development),b mediates immune cell adhesion,c and activates intracellular signalling.d,e This wide range of functional activities may seem surprising given hyaluronan's simple structure, but results in fact from the large number of hyaluronan-binding proteins (often termed hyaladherins) that exhibit significant differences in their tissue expression, cellular localization, specificity, affinity and regulation. Characterization of the molecular basis of hyaluronan recognition by proteins, and how this is modulated in vivo, will be a key determinant in understanding the biology of this important polysaccharide.
a See review by Hardingham in this series.
b See review by Toole in this series.
c See review by Strong and de la Motte in this series.
d See review by Knudson and Knudson in this series.
e See review by Turley and Harrison in this series.
Many hyaluronan-binding proteins contain a common domain, termed a Link module, which is involved in ligand binding (Figure 1). The Link module (also referred to as a proteoglycan tandem repeat)1 was first described in cartilage link protein, which is comprised of an immunoglobulin domain and two contiguous Link modules. This molecular arrangement is also found in the G1-domains of the chondroitin sulfate proteoglycans aggrecan, versican, neurocan and brevican. As shown in Figure 2, aggrecan forms huge link protein-stabilized complexes with hyaluronan that provide load-bearing function in articular cartilage.a Similar aggregates, in which versican, neurocan or brevican substitute for aggrecan, are likely to contribute to the structural integrity of many other tissues, e.g., skin, blood vessels and brain. The immunoglobulin domains are probably responsible for the link protein-proteoglycan interaction, while the Link modules mediate binding to hyaluronan.
a See review by Hardingham in this series.
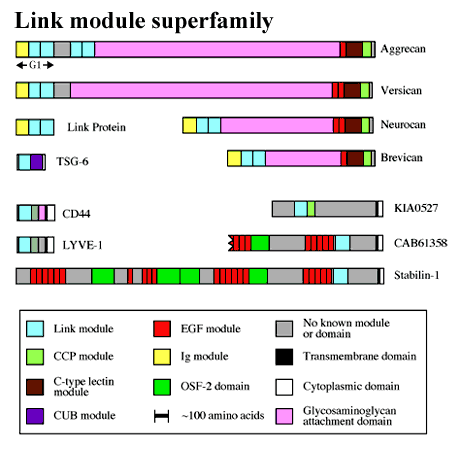
Figure 1 The Link module superfamily.
The ubiquitous hyaluronan receptor CD44 has diverse functions including the attachment, organization and turnover of extracellular matrix at the cell surface (e.g., in cartilage; Figure 2) and mediates the migration of lymphocytes during inflammation.d CD44 can exist in numerous isoforms, due to alternative splicing of 10 variant exons in different combinations, all of which contain a single Link module close to the N-terminus of the protein. Apart from CD44, the only other member of the Link module superfamily (Figure 1) that has been clearly identified as a hyaluronan receptor is LYVE-1, a recently discovered protein that is restricted in its expression to lymph vessel endothelium and that may be involved in hyaluronan catabolism.2
d See review by Knudson and Knudson in this series.
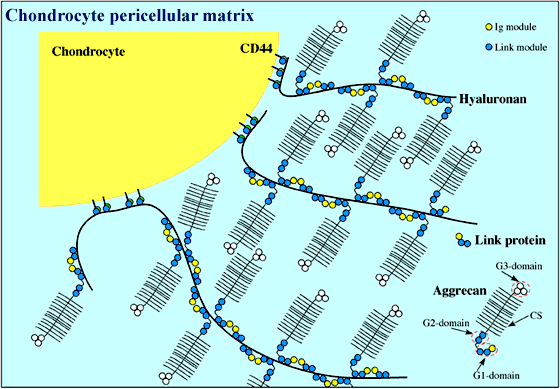
Figure 2
Chondrocyte extracellular matrix is composed of huge multi-molecular complexes consisting of an alternating array of link protein and aggrecan molecules along a central hyaluronan filament. These aggregates, some of which are anchored to the chondrocyte surface via the hyaluronan receptor CD44, provide cartilage with its load-bearing properties. Aggrecan is a proteoglycan made up of three globular domains (G1, G2 and G3) and a region where ~100 chondroitin sulfate (CS) chains are attached.
The protein product of tumor necrosis factor-stimulated gene-6 (TSG-6), which contains a single Link module, is secreted in response to inflammatory stimuli (e.g., in the articular joints of arthritis patients).3 TSG-6 has been implicated in the regulation of leukocyte migration, and the pattern of its expression suggests that it is likely to be involved in extracellular matrix remodeling. For example, TSG-6 in cartilage may inhibit the formation of hyaluronan-protein aggregates, promote aggregate dissociation or modulate CD44 function (Figure 3).4,5 In certain tissue locations (e.g., during the expansion of cumulus cell-ooycte complexes), TSG-6 can associate with the inter-a-inhibitor (IaI), and this TSG-6/IaI complex may be able to stabilize extracellular matrix by cross-linking of hyaluronan chains.f Although the role of TSG-6 protein is poorly understood, its hyaluronan-binding properties, and their structural bases, are probably the best characterized of any hyaladherin (see below).
f See Salustri and Fülöp in this series; O Carrette, R V Nemade, A J Day, A Brickner and W J Larsen, manuscript submitted.
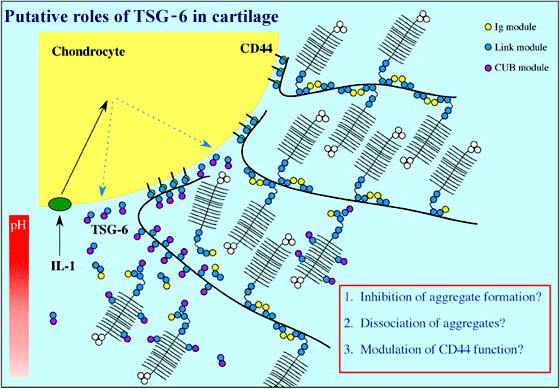
Figure 3 Putative roles of TSG-6 in cartilage.
TSG-6, which is secreted by chondrocytes during inflammation (e.g., in rheumatoid arthritis), is likely to be involved in extracellular matrix remodeling, as it can interact with components of cartilage such as hyaluronan, chondroitin-4-sulfate and the G1-domain of aggrecan.4,5 For instance, TSG-6 may inhibit the formation of cartilage aggregates, blocking their assembly by binding to the G1-domain and the chondroitin-4-sulfate chains of de novo synthesized aggrecan. It could also be involved in the dissociation of existing aggregates, possibly by altering the structure of hyaluronan, and the regulation of CD44 function. The hyaluronan- and aggrecan-binding activities of TSG-6 are pH-dependent; this may result in regulation of its functions via pH gradients that exist in articular cartilage. In this regard, there is likely to be a significant drop in pH in the peri-chondrocytic region during inflammation.4
Figure 1 shows several new members of the Link module superfamily (i.e., stabilin-1, CAB61358 and KIA0527) that are all likely to be cell surface proteins. Stabilin-1 (accession code AJ275213) and CAB61358 (which is the accession code for this incomplete sequence) have very similar domain organizations, and the Link modules of these proteins both show about 50% identity with that of TSG-6. However, it is unclear whether or not these are hyaladherins, as no data on their hyaluronan-binding properties are yet available. The same is true for KIA0527 (accession code AB011099), which was cloned from brain tissue.
As can be seen from Figure 4, there is a growing number of hyaluronan-binding proteins that do not contain a Link module. Most of these are unrelated to each other at the primary sequence level. There are few data on the positions of the hyaluronan-binding sites in these proteins, apart from the receptor RHAMM,e and therefore they will not be covered in this review.
e See review by Turley and Harrison in this series.
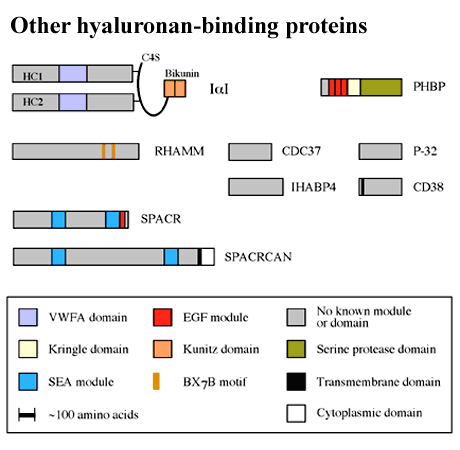
Figure 4 ther hyaluronan binding proteins.
The BX7B motif is shown only for RHAMM, for which there is evidence of its involvement in hyaluronan binding.
The Link module is a domain of approximately 100 amino acids with four cysteines disulfide-bonded in the pattern Cys1-Cys4 and Cys2-Cys3 (Figure 5). The three-dimensional structure of the Link module from human TSG-6 has been determined by nuclear magnetic resonance spectroscopy in solution.1 It is comprised of two a-helices and two triple-stranded anti-parallel ß-sheets (Figure 6). As can be seen from the alignment in Figure 5, many residues (including the four cysteines) are highly conserved across the Link module superfamily. There are no insertions or deletions present in regions corresponding to elements of secondary structure, apart from the bulge in the ß5 strand, and those that do occur would mainly affect the length of exposed loops. This structure therefore has defined the ‘consensus fold’ for the Link module superfamily as a whole. In practical terms, this means that the TSG-6 Link module co-ordinates can be used as a template for useful modeling of other superfamily members.6,7 A good test of this is the Link module from KIA0527, which despite having an unusual arrangement of cysteine residues (Figure 5), was successfully modeled, as shown in Figure 7.

Figure 5 Multiple sequence alignment of Link modules from the proteins in Figure 1.
LP, link protein; A, aggrecan; B, brevican; V, versican; STAB-1, stabilin-1; 61358, CAB61358; 0527, KIA0527. All the sequences are from human except for brevican which is from rat (R). Cysteine residues are shown in yellow and other highly conserved residues (i.e., identical in 2/3 of the sequences) are denoted in pink. These residues comprise the Link module consensus sequence (CON). The position of the secondary structure elements (SS), determined for TSG-6,1 are shown below the alignment (a = a-helix, b = ß-strand, ^ = bulge).
![]() enlarged Figure 5.
enlarged Figure 5.
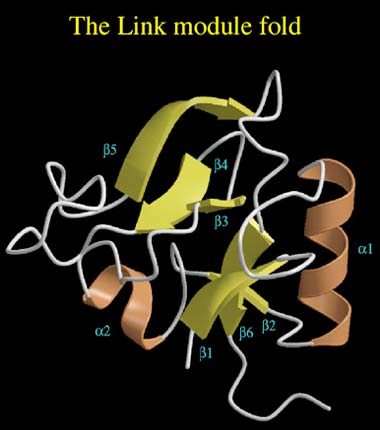
Figure 6 The Link module fold.
The structure consists of two a-helices (brown) and two triple-stranded ß-sheets (yellow). Sheet I is comprised of strands ß1, ß2, ß6, while sheet II is made up from strands ß3, ß4 and ß5.
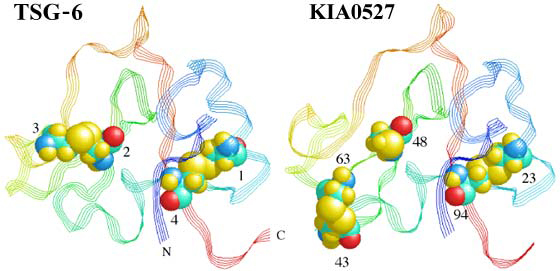
Figure 7 Link modules of TSG-6 and KIA0527.
The Link module of KIA0527 was modeled on the basis of the TSG-6 Link module co-ordinates1 and the pairwise alignment of these proteins from Figure 5 (A J Day, unpublished work). TSG-6 is a typical member of the Link module superfamily with disulfide bridges between consensus Cys1-Cys4 and Cys2-Cys3. In the model of KIA0527, consensus Cys-2 (residue 48) is a free sulfhydryl, due to consensus Cys-3 being absent, while the two additional cysteines (residues 43 and 63) are able to form a disulfide bond. The fact that this unusual arrangement of cysteines can be accommodated into the TSG-6 structure is a good indicator that this does represent the consensus fold for the Link module.
The recombinant domain used to solve the tertiary structure was found to be functionally active,1,4,5,8 indicating that in TSG-6 a single Link module is sufficient for high-affinity hyaluronan binding. This is not the case for CD44, which has a hyaluronan-binding domain of ~160 amino acids made up of a single Link module with additional N- and C-terminal extensions linked by a disulfide bond. These extensions are required for correct folding and functional activity (Figure 8).9,10 A larger domain still is likely to be utilized by link protein, aggrecan and the other aggrecan-like proteoglycans, as these proteins all have a pair of contiguous Link modules in their hyaluronan-binding regions (Figure 1). In link protein and aggrecan, both Link modules participate in ligand binding.11,12 Therefore, the proteins of the Link module superfamily can be divided on the basis of the size of their hyaluronan-binding domains into three sub-groups,7 as illustrated in Figures 9 and 10. The sizes of these domains appear to correlate broadly with the length of hyaluronan recognized. For example, hexasaccharides (HA6) and decasaccharides (HA10) are the minimum lengths of hyaluronan required for high-affinity binding to Type A and Type C domains, respectively.8 Hyaluronan binding to CD44 can be displaced by either HA6 or HA10, depending on the cell background on which it is expressed.13,14
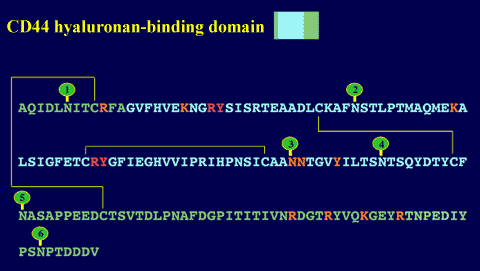
Figure 8 The CD44 hyaluronan-binding domain.
At ~160 amino acids in length, this domain consists of a Link module (blue) with N- and C-terminal extensions (green) that are linked by a disulfide bond. Residues that are essential or important for hyaluronan binding6,9 are denoted in red and orange, respectively. The positions of the six N-linked glycosylation sites are indicated by green lollipops.
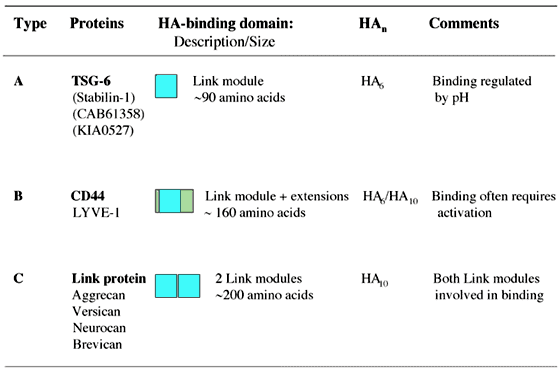
Figure 9 Link module sub-groups.
The Link module superfamily can be divided into three sub-groups (Type A, B and C) on the basis of the size of their hyaluronan-binding domains, which correlates broadly with the minimum size of hyaluronan (HA) recognized.7,8 Proteins highlighted in bold represent the archetypal member of each sub-group. LYVE-1 is likely to have a Type-B hyaluronan-binding domain as, like CD44, it contains an additional disulfide bond linking sequences flanking the Link module.2 The proteins shown in parentheses have not yet been demonstrated to be hyaluronan-binding proteins.
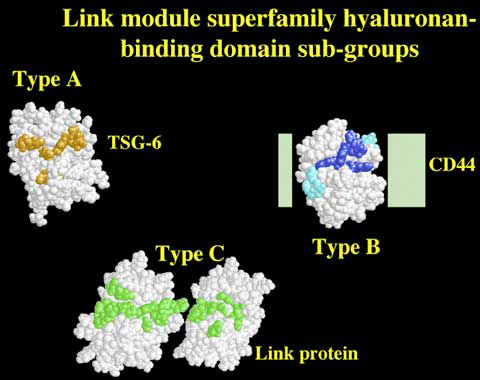
Figure 10 The structures/models of the Type A, B and C hyaluronan-binding domains of TSG-6, CD44 and link protein.
Only the tertiary structure of the Type A domain, a single Link module from TSG-6, has been determined to date. Residues that mediate the interaction with hyaluronan are colored in orange.g The Link modules shown for CD44 and link protein were modeled on the basis of the TSG-6 co-ordinates. In CD44 the structures of the N- and C-terminal extensions (light green boxes) are currently unknown. However, these are likely to form a sub-domain in intimate contact with the Link module.7 Amino acids of the CD44 Link module that are either critical or important for ligand binding are depicted in dark blue or light blue, respectively.6 In the Type C domain, the relative orientations of the two Link modules remain to be determined, and only one possible configuration is illustrated. The residues of link protein predicted to be involved in the interaction with hyaluronan are colored green (A J Day, unpublished).
Residues of CD44 involved in hyaluronan binding have been determined by site-directed mutagenesis.6,9 As can be seen from Figure 8, functionally important amino acids are present both on the Link module and on the flanking sequences. The ligand-binding residues (in the Link module) form a continuous surface on one face of the molecule (Figure 11). The same face of the Link module is involved in hyaluronan binding in TSG-6, indicating that the position of the ligand interaction surface may be conserved across the superfamily.8 From these studies, it is clear that in Link module-containing proteins, binding residues are brought together from different parts of the sequence and a folded structure is necessary to generate a ligand-binding surface (Figure 12). Linear sequence motifs (e.g., BX7B peptides found in RHAMM)e do not appear to be involved in Link module-hyaluronan interactions.8
e See review by Turley and Harrison in this series.
g DJ Mahoney and AJ Day, manuscript in preparation.
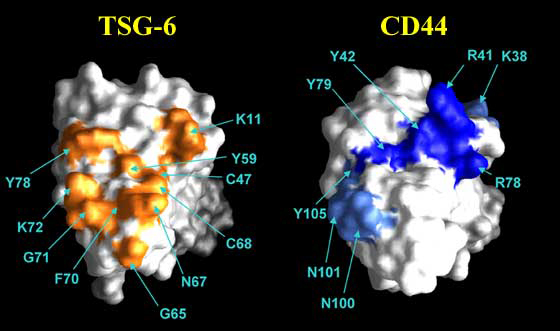
Figure 11 Hyaluronan-binding sites of TSG-6 and CD44.
The hyaluronan-binding sites of these proteins are on the same face of the Link module. Surface-exposed amino acids on the TSG-6 Link module that are perturbed on HA8 binding are shown in orange.8 Site-directed mutagenesis has allowed residues directly involved in the interaction with hyaluronan to be distinguished from those affected due to ligand-induced conformation changes.g Amino acids of the CD44 Link module that are either critical or important for ligand binding are depicted in dark blue or light blue, respectively.6
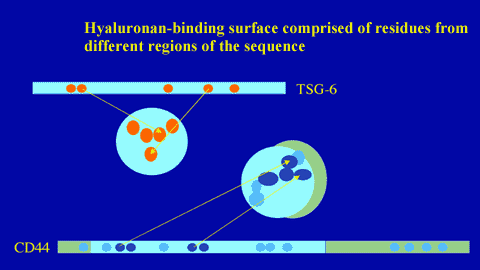
Figure 12
The hyaluronan-binding surface is comprised of residues brought together from different parts of the protein sequence.
Although, the hyaluronan-binding sites in TSG-6 and CD44 have similar locations on the Link module surface (Figure 11), there appear to be some major differences in the details of the residues and sequence positions involved in binding in the two proteins. Comparison of the Link module sequences of CD44 and TSG-6 with all the other members of the superfamily indicates that the molecular details of hyaluronan-binding (i.e., the interaction networks) are likely to be distinct in each protein. This is perhaps not surprising given the different sub-types of hyaluronan-binding domain (described above) as well as many other differences in specificity and regulation. For instance, TSG-6 binds to chondroitin-4-sulfate with high affinity, but this glycosaminoglycan is not recognized by aggrecan or link protein.5
Chemical modification studies have indicated that basic amino acids are major determinants in the binding of aggrecan and link protein to hyaluronan by forming ionic bonds with the carboxylic acid groups of the glycosaminoglycan.8 This is also likely to be the case for CD44, for which two arginines (Arg41 and Arg78) play a critical role in ligand binding.6,9 In addition, two tyrosines (Tyr42, Tyr79) are essential for CD44 functional activity (Figure 11); mutation of Tyr79 to phenylalanine destroys functional activity, indicating that its hydroxyl group makes a crucial hydrogen bond to the sugar.6 Other amino acids of CD44, including three lysines, four arginines, two asparagines and a tyrosine (shown in orange on Figure 8), are also implicated in hyaluronan binding but have a less critical involvement. The large number of residues engaged in binding indicates that an extensive network of interactions (particular to CD44) maintains its association with hyaluronan. The energetics of this are clearly finely balanced, as loss of a single hydrogen bond or ionic interaction can be enough to abolish binding. This is also true for TSG-6 in which five amino acids have been identified as having an important functional role, including basic and aromatic residues.g To date, the thermodynamics of hyaluronan-protein binding have been determined only for the TSG-6 Link module, in which the major energetic contribution driving the interaction is a large and exothermic enthalpy.8 The small change in heat capacity seen on binding is consistent with an intermolecular interface comprising a significant polar or charged component rather than the burial of a large hydrophobic surface area. While it is clear that ionic associations are important, non-ionic interactions (e.g., hydrogen bonding and van der Waals forces) are also likely to make a significant contribution to the binding energy.15 This is consistent with data from site-directed mutagenesis showing that in addition to a lysine, 3 tyrosines and a phenylalanine have a critical role in binding.g
g DJ Mahoney and AJ Day, manuscript in preparation.
Clusters of basic amino acids, referred to as BX7B motifs (where B is either lysine or arginine and X can be any amino acid apart from acidic residues), have been implicated in the interaction between hyaluronan and RHAMM.e Although BX7B-like sequences are present in many other hyaladherins (e.g., IaI, SPACR, SPACRCAN, CD38, CDC37, P-32 and IHABP4; Figure 4), there are no data to indicate that they mediate hyaluronan binding in these proteins. This spacing of basic amino acids is extremely common in protein sequences. Therefore, the presence of a BX7B motif should not be interpreted as an indicator that a protein will bind hyaluronan, nor should it be assumed that this is necessarily the site of ligand-binding activity in a hyaladherin.
e See review by Turley and Harrison in this series.
Unlike other members of the Link module superfamily, the interaction of TSG-6 with hyaluronan is highly pH-dependent (Figure 13), with maximal binding at pH 6 and a dramatic reduction in activity as the pH is increased.4 This may serve to regulate the ligand-binding functions of this protein in tissues that have significant pH gradients, for example, in inflamed cartilage where TSG-6 is expressed (Figure 3). The structural basis of this unusual pH-dependency is not yet fully understood. However, it is clear that no detectable alteration in the conformation of the TSG-6 Link module fold occurs between pH 6 and 8.15 One possible explanation is that raising the pH changes the charge-state on a histidine residue, which leads to the loss of a critical binding interaction.
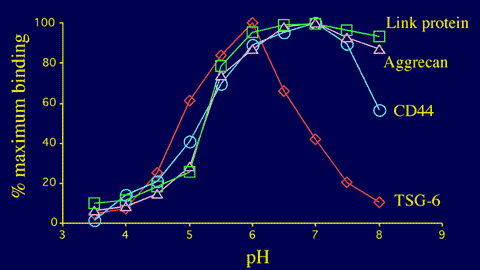
Figure 13 Effect of pH on the hyaluronan-binding activity of four members of the Link module superfamily.
The pH-dependency of the interaction between the TSG-6 Link module and hyaluronan is distinct from those of CD44 (A A Parkar, S Banerji, D G Jackson & A J Day, unpublished data), aggrecan and link protein.4
CD44 is present on many cell types (e.g., lymphocytes) in a non-functional form that requires some activation process to acquire hyaluronan-binding capabilities. The regulation of CD44 activity is a complex issue with many factors potentially involved. These include the cell background on which CD44 is expressed, post-translational modifications (e.g., glycosylation), the splice isoform involved, cytoskeletal attachment, molecular clustering/receptor density on the cell surface and phosphorylation of the intra-cytoplasmic domain.d,14,16 Many of these have already been reviewed in this series,d and therefore only the role of glycosylation will be discussed here. As shown in Figure 8, the hyaluronan-binding domain of human CD44 contains six potential sites for N-linked carbohydrate attachment. Expression of this region in E. coli in a non-glycosylated form10 and site-specific mutagenesis16 indicate that glycosylation is not required at any of these positions for either correct folding or functional activity. It is, however, one of the principal mechanisms modulating the interaction between CD44 and hyaluronan, and there is differential glycosylation of the receptor depending on cell type and cell activation state. Removal of N-glycans by mutation of the first or fifth glycosylation sites, which are on the sequences flanking the Link module, can switch CD44 on an "inducible" cell background into a constitutively active form.16 This may indicate that particular carbohydrate structures are able to obscure the hyaluronan-binding site. Another possibility is that certain N-glycans prevent receptor clustering or lock the protein in a non-binding conformation. The molecular basis of CD44 regulation should become clearer once the tertiary structure of the hyaluronan-binding domain is known. For instance, this will reveal the positions of the glycosylation sites relative to the hyaluronan interaction surface.
d See review by Knudson and Knudson in this series.
The Link module has a very similar fold to that of the C-type lectin domain.1 From Figure 14, it can be seen that these domains have identical topologies, with similar secondary structural elements and many residues in common stabilizing the hydrophobic core. Although the mode of carbohydrate binding is clearly distinct in these domains (i.e., the C-type lectin binds in a calcium-dependent manner, whilst the Link module-hyaluronan interaction is independent of metal ions), the positions of the ligand-binding surfaces are very similar.6 Both domains also mediate leukocyte rolling on the vascular endothelium during inflammation. Therefore, it seems very likely that they have a common evolutionary origin. The C-type lectin domain is present in many invertebrate proteins, whereas the Link module has only been identified in vertebrates, including sharks.17 This indicates that the Link module probably evolved from the C-type lectin domain sometime prior to the divergence of the cartilaginous fish and the bony fish/land vertebrates about 400 million years ago. Interestingly, Lee and Spicer18 have hypothesized that the ability to synthesize hyaluronan is a comparatively recent innovation in the evolution of metazoan organisms. For example, while a hyaluronan synthase has been found in the chordate Amphioxus, there are no such enzymes encoded in the Drosophila genome.h Therefore, it is possible that the Link module and hyaluronan appeared at approximately the same time during evolution.
h See review by Spicer and McDonald in this series.
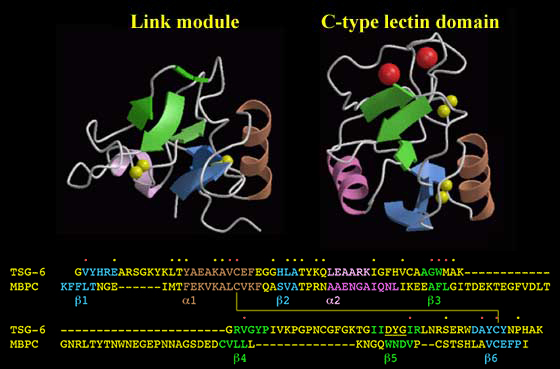
Figure 14 The Link module and the C-type lectin domain.
These modules have identical topologies and a very similar organization of secondary structure. An alignment of the Link module from human TSG-6 with the C-type lectin domain from human mannose-binding protein C-chain (MBPC) shows that the secondary structural elements are found in equivalent sequence positions. The yellow/red dots denote the positions of amino acid identities and conservative replacements. The hydrophobic interiors of the two domains are similar, with core residues in the C-type lectin domain found at equivalent sequence positions to those in the Link module (red dots). This includes consensus cysteines Cys1 and Cys4, which are linked by disulfide bonds in each module. The major difference between the structures is the length of two loops. The Link module has a short loop connecting the ß3 and ß4 strands, whereas in the C-type lectin domain, there is a region of ~35 amino acids containing residues responsible for chelating two Ca2+ ions (large red spheres). Similarly, the 14-residue loop between the ß4 and ß5 strands in the Link module is shorter in the lectin domain. The ß5 strand of the Link module is shown in two parts as it is interrupted by a bulge (underlined) as illustrated in Figure 5.
Significant advances have been made in the last few years in our understanding of hyaluronan-protein interactions. However, this knowledge is far from complete, and further studies to determine the three-dimensional structures of different types of hyaluronan-binding domains, and their ligand complexes, are clearly essential if we are to unravel the molecular mechanisms underlying the biology of this amazing polysaccharide.