
Hidenori Otsuka
Professor, Department of Applied Chemistry, Faculty of Science, Tokyo University of Science (TUS)
He received his Ph.D. (Science) from Tokyo University of Science. Thereafter, he moved to Ciba-Geigy Japan Co., Ltd., now Novartis Japan in 1995. He was recipient of JSPS (Japan Society for the Promotion of Science) Research Fellowship for Young Scientists at the University of Tokyo Graduate School and Tokyo Women's Medical University in 1997-2001. Thereafter, he was a Researcher at the National Institute of Advanced Industrial Science and Technology (AIST), and Principal Researcher at the National Institute for Materials Science (NIMS). He became an associate professor in 2006 and currently serves as a professor in the Faculty of Science and the Graduate School of Science at TUS.
Major honors include The Award for Young Investigator of Japanese Society for Biomaterials (2005) and Iketani Science and Technology Foundation (2022). He has been recognized as a Member of Sigma Xi and National Council Member of the International Association of Advanced Materials (IAAM).
His research has focused on biofunctional interfacial material development utilizing interfacial colloid chemistry—Synthesis of blood compatible polymer surfaces, hydrogels for targeted drug delivery systems (DDS) and tissue engineering.
As a tissue engineering approach in regenerative medicine, we developed a novel hydrogel to serve as a scaffold for culturing cells in three dimensions and established a method for its simple synthesis. Here, hydrogel refers to a polymer chain network that contains water or other liquids. In vivo, cells proliferate and differentiate using the extracellular matrix (ECM), formed from various biomacromolecules such as collagen and hyaluronic acid, as a scaffold. Therefore, when culturing cells in vitro, a material serving as a scaffold is also required. Recently, a hydrogel called an interpenetrating polymer network (IPN), where multiple polymers intertwine to form independent, multiple network structures, has gained attention as a material mimicking the ECM.
Chitosan (CH) is widely used as a biomaterial due to its high biocompatibility and biodegradability. Therefore, in this study, we developed an interpenetrating polymer network hydrogel (hereafter referred to as IPN gel) composed of CH, polyethylene glycol (PEG), and self-assembling peptides as a cellular scaffold material. Specifically, this technology enables the simple, in-situ, one-pot synthesis of the IPN network. This is achieved through the phenomenon of self-organization, where the peptide autonomously forms a fibrous β-sheet structure, followed by the formation of covalent bonds between CH and PEG. Indeed, the developed IPN gel possesses a structure suitable for a scaffold, and it was confirmed that articular chondrocytes actively proliferate and regenerate in the scaffold’s IPN.
Articular cartilage is an extremely thin tissue, approximately 2 to 7 mm thick, yet it plays a crucial role in joint function. Its histological composition is highly specialized, with chondrocytes constituting only about 2% of the abundant cartilage matrix. Furthermore, chondrocytes have low proliferative capacity and are not highly active. Consequently, once articular cartilage is damaged, the mechanisms for repairing the injury are difficult to activate, and it has limited self-repair capabilities. Currently, there are no effective treatment methods for cartilage defects caused by trauma, osteochondritis dissecans, or osteoarthritis.
Tissue engineering holds great promise as an approach to address this issue. While autologous cartilage transplantation and autologous cell transplantation have been performed to date, an unavoidable problem is the age of the donor from whom the patient's own tissue is obtained. Particularly in treating joint disorders, many patients are elderly, presenting challenges such as age-related limitations in cartilage matrix synthesis and a decrease in the molecular weight of hyaluronic acid, a key component of this matrix. The novel cell scaffold material synthesis technology developed in this research is expected to play a crucial role in advancing cartilage regenerative medicine in today's rapidly aging society. Furthermore, the absence of immune rejection risk makes this a highly promising medical technology with significant demand.
Articular cartilage is classified as hyaline cartilage, with a composition of approximately 80% water, 12% collagen, 2% proteoglycans and hyaluronic acid, and about 6% other components. Chondrocytes constitute only 2-3% of the cartilage volume. When comparing the proportion of total protein on a dry weight basis, type II collagen is estimated at about 60% and aggrecan at about 35%.
The cartilage matrix consists of fibrous components made of collagen, proteoglycans such as hyaluronan and aggrecan that fill the interstitial spaces, and other glycoproteins. While the former fibrous components (made of collagen; the first network) form the basic structural framework of the tissue and provide rigidity, the latter fibrous components (mainly aggrecan and hyaluronic acid; the second network) deposit between the fibers, giving cartilage its characteristic hyaline appearance and elasticity. These molecules, aggrecan and hyaluronic acid, together with link proteins (LP), form distinct structures called proteoglycan aggregates that deposit within the tissue (Fig. 1)1,2.
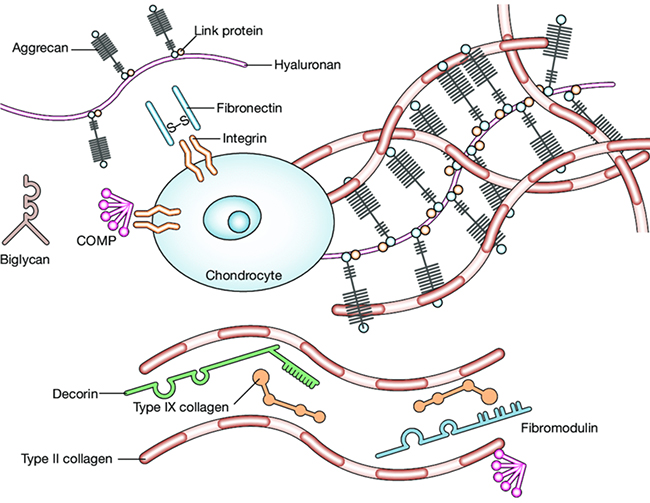
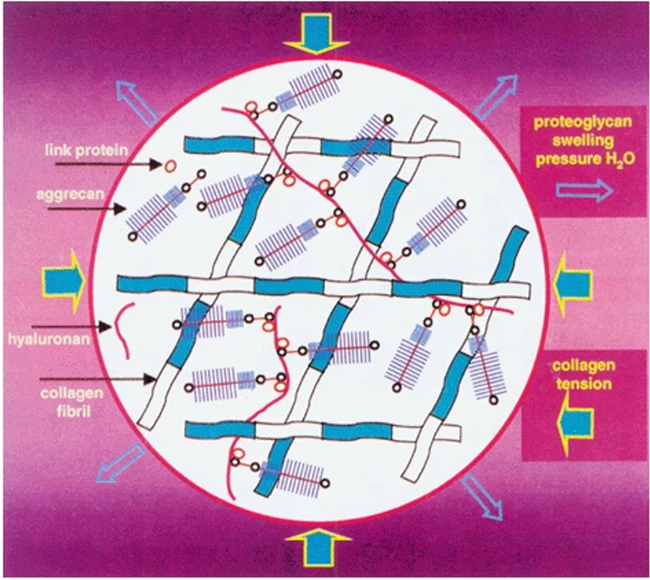
At high concentrations, proteoglycans create a large osmotic swelling pressure and draw water into the tissue. This occurs because all of the negatively charged anionic groups on the glycosaminoglycan (GAG) chains of aggrecan carry with them mobile counter ions such as Na+. This creates a large difference in ion concentration between the cartilage and surrounding tissue and an imbalance amongst the freely diffusible anions and cations. Water is drawn into cartilage because of this osmotic imbalance and because aggrecan is too large and immobile to redistribute itself. The addition of water causes the aggrecan-rich matrix network to swell and expand. This water-swollen matrix is critical to the biomechanical properties of cartilage. Another feature of the composite collagen/aggrecan organization is also important: the collagen/aggrecan network is stiff and resistant to deformation. In other words, the collagen meshwork structure significantly restricts the mobility of aggrecan within the matrix, resulting in the generation of expansion pressure. Aggrecan also offers great resistance to any fluid flow and redistribution of water. Thus, cartilage is referred to as a visco-elastic tissue in that it behaves like a stiff elastic polymer that is resistant to sudden impact loading, yet shows some slow inelastic deformation with sustained loads (Fig. 2)3.
The ECM that constitutes our bodies is formed from a diverse network of biomacromolecules. When attempting to regenerate defective tissue, transplanting cells together with materials that mimic the ECM structure within the body is expected to efficiently regenerate tissue that retains the structural and functional characteristics of the target organ. Therefore, the development of cell scaffolds that reproduce the ECM structure, particularly hydrogels, is urgently necessary for tissue regeneration. Hydrogels engineered with biologically derived macromolecules such as collagen and hyaluronic acid—key components of the ECM—exhibit high biological activity but suffer from insufficient mechanical strength. To enhance both in vivo degradation kinetics and mechanical properties, we investigated the incorporation of a secondary network into the gel matrix, this notion having been inspired by the composite network formed by collagen and hyaluronic acid found in biological tissues.
IPN is a material in which heterogeneous polymer networks exist independently of each other, enabling precise control of the biological and physical properties of hydrogels and synergistic functional enhancement as a structured material4-6. The fusion of synthetic materials that compensate for the shortcomings of biologically derived materials while retaining their functions can be achieved either through physical entanglement or covalent crosslinking. However, the key issue that arises here is “how to construct this network structure.” The conventional method for preparing IPN gels typically requires an external stimulus, such as light irradiation or the addition of other chemicals, to form the second network after the first network has been established. This method of network formation requires a multi-step process and generates cytotoxic radical species, making it unsuitable for constructing scaffold materials for tissue regeneration. Therefore, for medical applications, particularly in tissue engineering, it is ideal to create replacement materials simply by injecting them directly into the defective site along with cells. If IPN gel could be spontaneously formed simply by injection, it would maintain a high quality of life post-treatment and, above all, enable the realization of minimally invasive therapy capable of efficiently regenerating cartilage tissue. In other words, the critical focus in scaffold material fabrication lies in creating an IPN structure mimicking the in vivo structure and using it in an injectable form without external stimulation.
The rationale for developing injectable IPN gels lies in “enabling the structuring of biodegradable IPN gels in injectable systems by strategically utilizing the ‘difference in crosslinking driving force’ and ‘time lag in gelation rate’ between heterogeneous networks.” Therefore, we selected two networks: a self-assembling peptide (RADA16) and chitosan (CH)-polyethylene glycol (PEG) (CH/PEG). RADA16 is a self-assembling peptide that physically forms an injectable gel. Upon salt addition or pH change, it simultaneously induces electrostatic and hydrophobic interactions between molecules, immediately forming a β-sheet structure (Fig. 3)7,8.
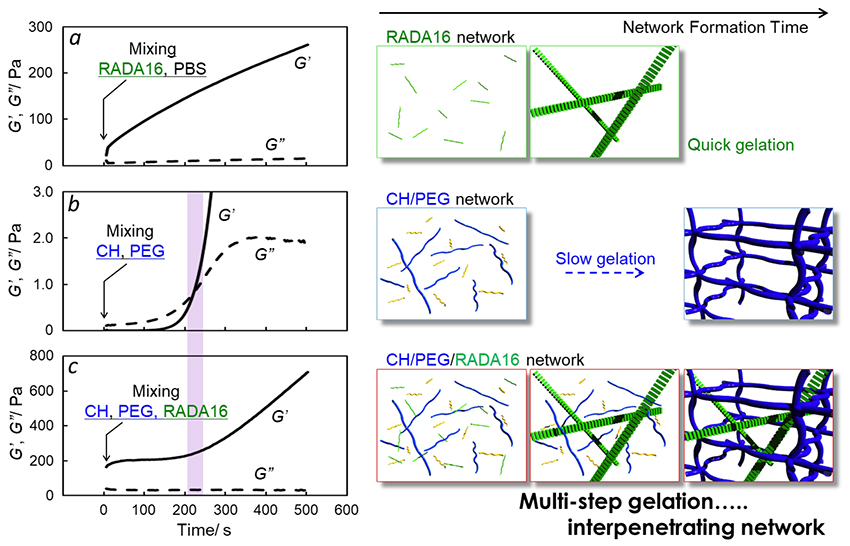
The CH forming the other network dissolves readily under physiological conditions and possesses abundant amino groups. Utilizing the properties of this CH, cross-linking reactions proceed based on the formation of amide bonds between both-terminally succinimidyl PEG (NHS-PEG-NHS) and CH9,10. The gelation in this case is not immediate; rather, it is an injectable gel that undergoes a slow phase transition from a viscous sol to a viscoelastic gel after a certain period of time. In this manner, considering the “gelation principle” and “gelation rate” in both networks, we inferred that mixing these gel precursors would enable the one-pot preparation of an injectable IPN gel based on the spontaneous sequential crosslinking of the two networks. To demonstrate this, the gelation rate was investigated via viscoelastic measurements. For the RADA16 peptide, immediately after mixing with phosphate buffered saline (PBS), the storage modulus was higher than the loss modulus, indicating gelation (first network formation) was extremely rapid. In contrast, for the CH/PEG gel, a sharp increase in storage modulus was observed after a predetermined time following solution mixing, confirming a distinct gelation point and indicating slower gelation (second network formation) compared to RADA16. In the gelation profile of the blended CH/PEG/RADA system, the storage modulus underwent multimodal changes, and the timescale of its inflection point showed a high correlation with the gelation point of the CH/PEG gel. These results explain the sequential formation of the network structure (Fig. 4)11.
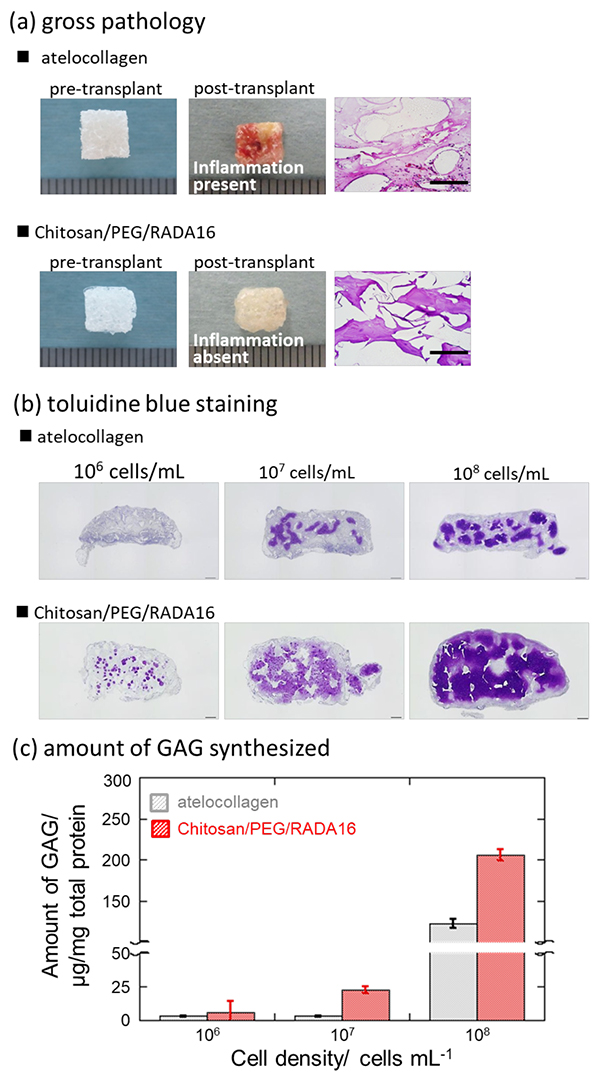
Functionally, it provided a cell scaffold that synergistically enhanced the tissue differentiation potential of the first network and the high strength of the second network. The efficacy of this IPN gel was also confirmed in in vivo transplantation experiments (Fig. 5)11. Gels containing human-derived chondrocytes at different cell densities were injected subcutaneously into 6-week-old mice and cultured for 8 weeks. Macroscopic pathological findings revealed a distinct inflammatory response in the atelocollagen gel post-transplantation, whereas no inflammation was observed in the IPN gel. Additionally, cartilage matrix formation was confirmed using toluidine blue (TB) staining. In TB staining, acidic mucopolysaccharides present in connective tissue (hyaluronic acid) and cartilage matrix (sulfated mucoprotein, chondroitin sulfate), when bound to the basic tar-based dye TB, stain in metachromatic fashion, i.e., exhibit metachromasia. The results revealed that IPN gel demonstrated superior chondrocyte differentiation in the production of hyaluronic acid and GAGs with fewer cells, significantly promoting effective regeneration of human cartilage.
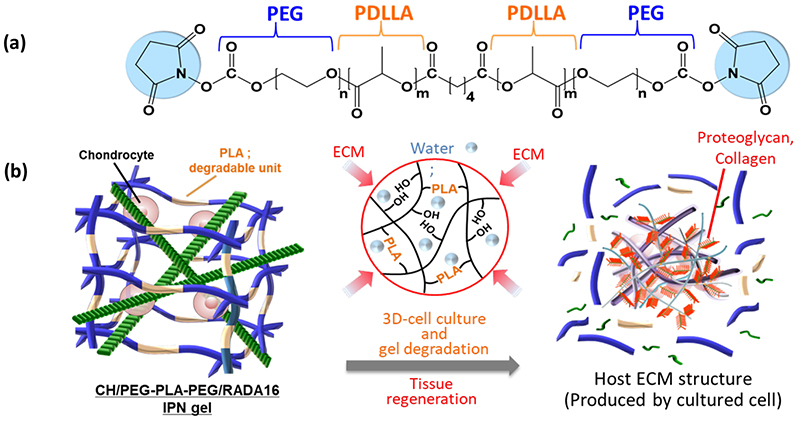
Hydrogel scaffolds designed for tissue regeneration ideally function as temporary containers for the ECM produced by cells embedded within the gel, ultimately being replaced by biologically derived tissue as the hydrogel degrades. If we can impart favorable degradation properties to this IPN gel, the synthetic IPN scaffold material is expected to be replaced by the ECM produced by cells, thereby promoting tissue regeneration. In this study, we newly prepared a biodegradable IPN gel using PEG-polylactide (PLA)-PEG (Mn ≈ 5K) as the crosslinker, which incorporates poly(D,L-lactide) (PDLLA) as a biodegradable molecule, replacing the conventional PEG crosslinker (Fig. 6)12.
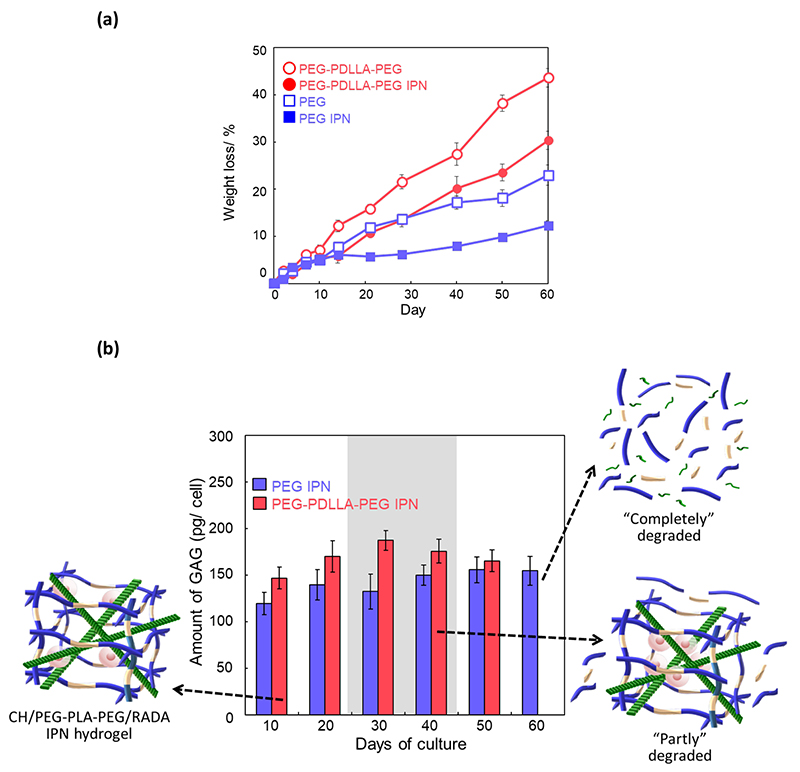
Focusing on hydrolysis originating from the ester bonds of PDLLA, we investigated the incorporation of PLA units into PEG crosslinkers and the resulting gel degradation behavior. Fig. 7a shows the degradation behavior of the gel over time when immersed in 150 mM PBS under physiological conditions (37°C, 5% CO2). Here, PEG (Mn ≈ 5K, Fig. 6) was used as a non-degradable crosslinker. The degradation rate of the gel using PEG-PLA-PEG as the crosslinker accelerated by approximately 2-3 times compared to the gel using a non-degradable crosslinker. For both crosslinkers, a delay in degradation behavior dependent on network reinforcement via IPN formation with the RADA16 peptide was observed, resulting in gradual gel degradation behavior over 8 weeks.
When aiming for tissue regeneration, the cell encapsulation strategy in hydrogel scaffolds involves mixing cells with precursors in a liquid solution, followed by gelation and cell encapsulation. Since the cross-linked structure greatly influences the diffusion of glycosaminoglycans and collagen, which are major components of the newly synthesized ECM by cells, scaffold degradation must closely follow ECM synthesis and macroscopic tissue development. Gel precursors, gelation mechanisms, and degradation products must possess cell compatibility. As described in “Molecular Mechanisms of Cartilage,” the load-bearing capacity of cartilage is maintained by two forces: the swelling pressure resulting from the high water-holding capacity and osmotic expansion pressure of GAGs, and the swelling pressure generated by collagen fibers compressing and restricting this expansion. Structurally, collagen fibers and proteoglycan molecules form an IPN structure, imparting compressive strength to cartilage. Fig. 7b shows the time-dependent changes in GAG accumulation within the gel during three-dimensional culture of bovine primary chondrocytes. GAG accumulation in the gel using a degradable crosslinker increased as the gel degraded and was superior to that in the gel using a non-degradable crosslinker. Ultimately, the gel using the degradable crosslinker degraded in approximately 7 weeks, but the GAG accumulation effect increased as the gel degraded. Combined with the results showing enhanced degradability of the biodegradable IPN in Fig. 7a, these results suggest that the biodegradable IPN gel successfully replaced the production matrix, indicating the construction of an ideal tissue regeneration scaffold. Furthermore, when using a degradable crosslinker, compared to a non-degradable crosslinker, the expression of the col2a1/ACAN/SOX9 gene cluster—a factor suggesting hyaline cartilage formation constituting articular cartilage—was enhanced. Moreover, TB staining shown in Fig. 5c revealed accumulation of acidic mucopolysaccharides such as hyaluronic acid and GAGs. This suggests that the application of degradable IPN to articular cartilage regeneration not only efficiently promotes cartilage ECM production but also achieves tissue regeneration closer in structure to that of actual articular cartilage tissue. Thus, biodegradable IPN is expected to be applied as a scaffold that undergoes progressive replacement over time with the ECM produced during tissue regeneration.
This IPN gel allows easy control of degradation rate and mechanical strength by adjusting the mixing ratio of degradable and non-degradable crosslinking agents. Consequently, it enables controlled release of encapsulated drugs and, furthermore, facilitates the construction of optimal environments tailored to the target tissue—not only for regenerating articular cartilage tissue but also for bone, skin, nerves, and other tissues. This makes it readily applicable to multi-organ tissue regeneration. Furthermore, the conventional IPN gel preparation requires a multi-step synthesis process. These IPN gels necessitate the formation of multiple networks independently, demanding multiple operations. Furthermore, the formation of the secondary network involves the generation of radical species that cause cytotoxicity and genetic damage, making it unsuitable for clinical application. However, this IPN gel can be prepared in a one-pot process without requiring special external stimuli, yielding an injectable gel with a biodegradable IPN structure. This provides excellent potential for development into clinical applications, such as articular cartilage tissue regeneration.
Furthermore, it enables superior tissue regeneration simply through the gel properties of the culture environment itself, without requiring protein-based pharmaceuticals that could potentially trigger an immune response (toxicity). Compared to existing scaffold materials like atelocollagen, it offers higher safety (no immune response) and allows for simpler scaffold material fabrication. In other words, it is expected to become a versatile scaffold material replacing conventional cell transplantation materials, paving the way for significant progress in regenerative medicine.