
Atsushi Miyagawa
Associate Professor, Department of Life Science and Applied Chemistry, Nagoya Institute of Technology
After graduating from the Department of Functional Materials Science, Faculty of Engineering, Saitama University in 2001, Atsushi Miyagawa completed his Ph.D. from the Department of Chemistry and Biotechnology, The University of Tokyo in 2006 under the supervision of Prof. Kenichi Hatanaka. Then, he became a postdoctoral fellow in the Synthetic Cellular Chemistry Laboratory, RIKEN, led by Prof. Yukishige Ito. He was appointed assistant professor at the Department of Life and Materials Engineering, Nagoya Institute of Technology in 2009, and then promoted to associate professor in 2019.
Polysaccharides consist of a sequence of monosaccharides connected through one or more types of bonds between saccharide units. These polysaccharides are not only known as energy storage molecules or cell wall structural components, but also as bioactive molecules with diverse biological regulatory functions. However, due to their structural complexity, it remains difficult to clarify the detailed functions of polysaccharides. To address this problem, functional analysis with chemically synthesized partial structures has been actively pursued. Among these efforts, attempts have been made to mimic polysaccharide structures by systematically placing monosaccharides or oligosaccharides in polymers.
This article focuses on β-glucans, a class of polysaccharides known for their immunostimulating effects and their long-standing use as pharmaceuticals. We mainly present our research findings on the chemical synthesis of β-glucans and the use of polymerization techniques to construct mimetic molecules.
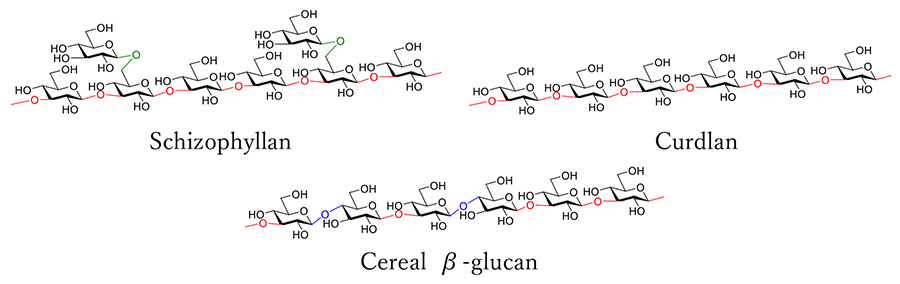
β-Glucans are a class of polysaccharides composed of glucose units linked via β-1,3 glycosidic bonds. Some β-glucans also include branched chains with β-1,6 linkages at the branching points or have β-1,4 linkages in their main chains (Figure 1)1-3. β-Glucans are widely distributed in various organisms, including bacteria, fungi, algae, and plants, where they serve as major structural components of cell walls or as energy sources. Among β-glucans derived from fungi, lentinan from Lentinula edodes and schizophyllan from Ganoderma lucidum have attracted significant attention and have been used as immunostimulatory pharmaceutical agents known for their antitumor activity.
In contrast, curdlan, a β-1,3-linked glucan without branching, is not soluble in water and has been studied for its unique helical structure and gel-forming properties4. Additionally, β-glucans with β-1,4 linkages in their main chains are abundant in cereal grains such as barley and oats and have also been reported to be immunostimulatory. Furthermore, β-glucans have a wide range of physiological activities, including antimicrobial, antioxidant, gut microbiota-modulating, blood glucose-regulating, cholesterol-lowering, and anti-inflammatory effects5-7.
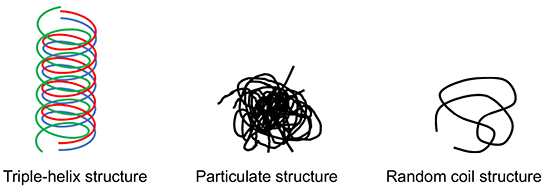
One of the characteristic three-dimensional structures of β-glucans is the triple-helix structure. Additionally, aggregated particulate structures and random coil conformations have also been reported8,9. The triple-helix structure is stabilized by intramolecular and intermolecular hydrogen bonds that can be disrupted in strongly alkaline aqueous solutions or in dimethyl sulfoxide, leading to the unraveling of the structure into single-stranded random coils. On the other hand, highly hydrophobic β-glucans exhibit low water solubility and tend to form aggregated particulate structures. These particulate forms are commonly observed in commercially available β-glucans used in health supplements.
Among these structures, the triple-helix conformation has attracted special attention due to its ability to form complexes with DNA and hydrophobic compounds (folic acid and polyphenol), thus facilitating its application as a delivery vehicle. In addition, the triple-helix structure has been proposed as a possible mechanism underlying the immunostimulatory effects of β-glucans10. Concurrently, studies on particulate β-glucans have also progressed, and their immunostimulatory activities have been reported11. Furthermore, β-glucans with a molecular weight of approximately one million are known to exhibit immunostimulating effects, while those with significantly lower molecular weights, on the order of thousands, function as an antagonists to immunostimulation. This finding highlights the importance of molecular weight in determining the immunological activity of β-glucans12.
Despite extensive research, the relationship between the three-dimensional structure of β-glucans and their immunostimulatory effects remains unclear. Since the different structures of β-glucans have been investigated separately, it remains difficult to compare the properties of these structures directly.
For the methods of β-glucan production, detailed descriptions of approaches involving the use of fungi or enzymes can be found in previous reviews13,14. This section focuses on the recent advances in chemical synthesis methods, especially those reported after 2019, while previous studies have been reviewed elsewhere15.
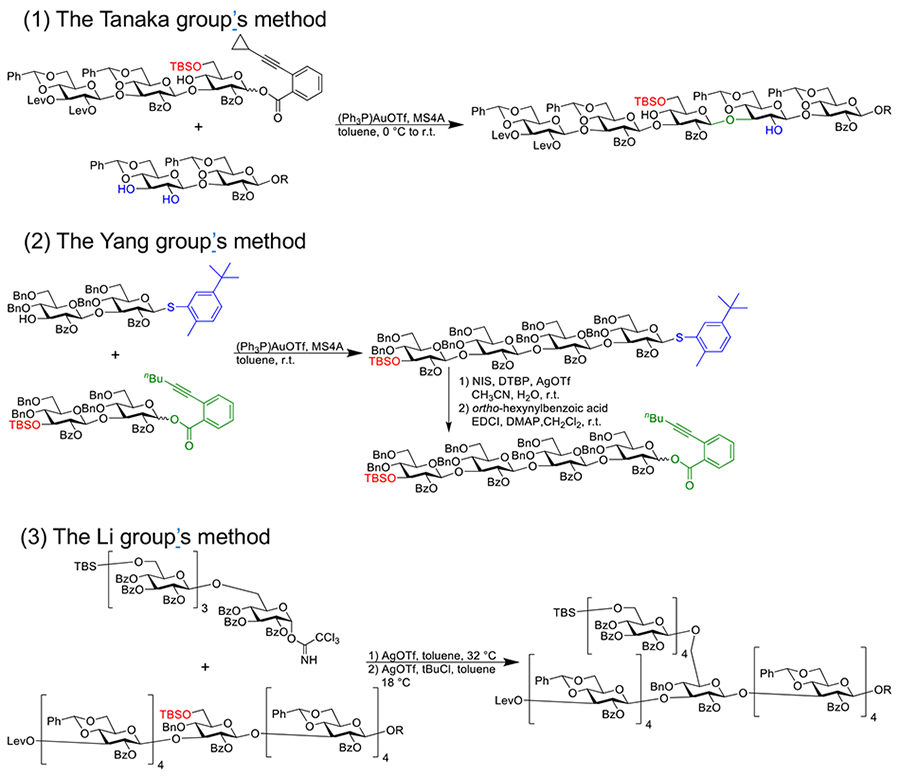
In 2019, Tanaka et al. reported the synthesis of a schizophyllan-type β-glucan composed of 14 glucose residues16. Based on their previous studies, this synthesis used a regioselective glycosylation method, which utilized the sugar acceptor with unprotected 2,3-hydroxyl groups (Figure 3 (1)). The glycosylation using trisaccharide building blocks was conducted under mild conditions using gold catalysts since the benzylidene group contained in the building blocks was sensitive to acids. For the introduction of branching chains, the benzylidene group was reductively cleaved, followed by silyl protection. After the construction of the main chain was completed, the branch chains were simultaneously introduced to positions that were deprotected by removal of protective silyl groups. Yang et al. synthesized a 16 mer linear β-glucan through a convergent approach, using armed acceptors and donors with benzyl groups that protect the 4,6-hydroxyl groups (Figure 3 (2))17. The donor's anomer position was activated by an alkynyl benzoate group, while the acceptor's anomeric position was protected by a sterically bulky 5-tert-butyl-2-methylbenzenethionyl group. After glycosylation, the anomer position in the thioglycoside is replaced by the alkynyl benzoate to convert it to a donor.
Li et al. synthesized a β-glucan with a linear backbone of nine glucose residues and branched chains of four glucose residues, connected via β-1,6 linkages. Their approach consisted first of synthesizing a tetrasaccharide building block for the main chain, followed by adding monosaccharides as branch units. Then, the pentasaccharide was glycosylated with the tetrasaccharide building block to construct a nonasaccharide and subsequent assembly into the final structure by introducing a branch unit (Figure 3 (3))18.
In construction of β-1,3 linkages, many researchers used a 4,6-benzylidene group as a protective group, with acetyl or benzoyl groups at 2-position to enhance β-selectivity. However, to control the reactivity of the 3-hydroxyl group, careful selection of protective groups is required to minimize the steric hindrance of the 4-position hydroxyl group and to introduce armed protecting groups. The benzylidene group, as a rigid bridging protection group, is advantageous but poses challenges to introduction of branched chains. To solve this problem, the benzylidene group is reductively cleaved to produce a structure with a benzyl group at the 4-position and an unprotected hydroxyl group at the 6-position, where selectively removable functional groups such as silyl or carbonate groups are introduced.
Our research group has developed a synthetic approach to inserting branching chains using a temporary benzylidene group19. In this method, we synthesized donors and acceptors with cleaved benzylidene group and employed chloroacetyl groups at the 6-position to distinguish this unit from acetylated units. After the assembly of a trisaccharide backbone, the chloroacetyl group was selectively removed to enable the introduction of branching chains at the positions. This process facilitated the synthesis of branching oligosaccharides, which led to the design of schizophyllan mimetics using β-glucan oligosaccharides with one branch at the central position.
The polymerization for clustering sugar units has been effective as a strategy to enhance the weak interactions between carbohydrate-recognizing proteins and sugar molecules. When carbohydrate-recognizing proteins have multiple carbohydrate-binding sites, the simultaneous binding of multiple sugar units can cause a clustering effect that significantly increases binding affinity for the proteins20–22. This approach has been particularly successful in studies of high-affinity polymers targeting plant lectins and bacterial toxins.
However, as far as I know, research on the binding between polysaccharide mimics and their protein binding partners has been limited. This is because the synthesis of sugar units recognized by polysaccharides is challenging and the subsequent polymerization of these sugar units is even more difficult. For polysaccharides with ionic functional groups such as amino, sulfate, or carboxy groups, polymers with strong interactions have been synthesized by replicating these ionic bonds. It has also been reported that attempts have been made to achieve strong interactions by mimicking the repeated structures of polysaccharides through polymerization of disaccharide or monosaccharide units23,24.
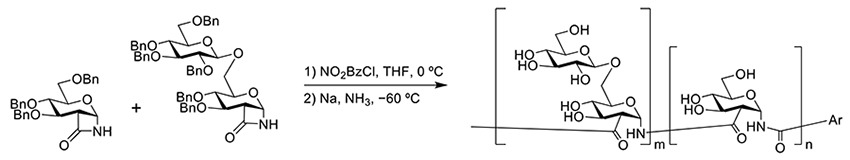
In contrast, studies on neutral polysaccharide mimics without ionic functional groups have not been reported much. Among them, a recent example of β-glucan mimetic polymers has been reported. Grinstaff’s group synthesized β-glucan mimetics by ring-opening polymerization using glucose lactam and gentiobiose lactam (Figure 4)25. The main chain consists of amide bonds, and although the β-glucan mimic lacks β-1,3 linkages, its linear arrangement of glucose units has branches of β-1,6-linked glucose derived from gentiobiose. Circular dichroism (CD) spectroscopy confirmed that these mimic molecules adopt an α-helical structure in aqueous solutions. The biological activity of these mimic molecules was reported to activate immune responses via Dectin-1. A polymer containing approximately 30% gentiobiose exhibited the highest activity, and the activity increased with a higher molecular weight. These mimic molecules replicate the α-helix structure of β-glucans, with the β-1,6-linked glucose lying exposed on the outside of the helix structure, highlighting the importance of branched chains.
Meanwhile, we have developed the synthesis of β-glucan mimics that clustered the β-glucan ligands. Previous studies suggest that approximately 10 sugar residues are required for recognition of the β-glucan by its receptor, Dectin-1, although oligosaccharides with only three or six sugar residues have also been reported to act as ligands26–28.
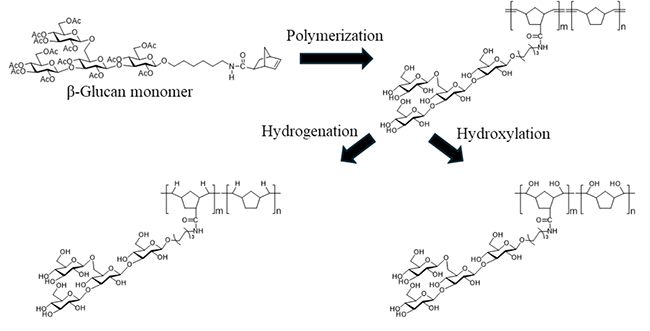
Based on studies showing that the minimum binding unit of Dectin-1 is a linear trisaccharide and that branched β-glucans have higher immunostimulatory activity, we designed a tetrasaccharide consisting of one branched sugar unit at the center of a linear trisaccharide. This was proposed as the minimum unit for binding to Dectin-1. Given the diverse properties of β-glucans in solution, we planned to modify the backbone of the polymer after polymerization to prepare polymers with different properties.
Specifically, in order to mimic the hydrophobic properties of β-glucans, we used norbornene, a hydrophobic backbone monomer, for polymerization and synthesized polymers of β-glucan tetrasaccharides. For comparison, polymers of linear trisaccharides were synthesized, as well as polymers with different sugar density. The double bonds formed during polymerization were then hydrogenated and hydroxylated to modify the polymer backbone (Figure 5).
As a result, we constructed a library of β-glucan mimic polymers with identical molecular weights and polydispersity and different backbone structure. This library, which includes different ligand structures, sugar content, and backbone structures, serves as a valuable tool for detailed study of interactions with sugar-recognizing proteins.
Tanaka et al. reported that even synthesized β-glucans with sufficient chain lengths to bind to Dectin-1 do not necessarily induce a strong immune response29. In contrast, studies using natural β-glucans have indicated that the clustering of multiple Dectin-1 receptors is essential for effective immune activation30. These results suggest that high molecular weight plays a crucial role in the immune response, leading to the adoption of a polymer-based approach.
Using RAW264 cells derived from mouse macrophages, we evaluated the immune activation potential of our polymer library. Figure 6 summarizes the ELISA test results of the relative evaluation of TNF-α, IL-1β, and IL-6 production as indicators of the immune response31. It presents the results for the polymer with a sugar-norbornene ratio of 1:3, which exhibited the highest activity. Natural Schizophyllan (SPG) was used as the reference material. Our results showed that polymers containing branched tetrasaccharides, especially those with unmodified or hydrogenated backbones, had significantly high activity. While polymers containing linear trisaccharides with similar structures showed comparable activities to SPG. This result demonstrated that the presence of branched chains is crucial for inducing strong immune responses. In addition, the hydrophobic backbone was identified as another contributing factor to immune activation.
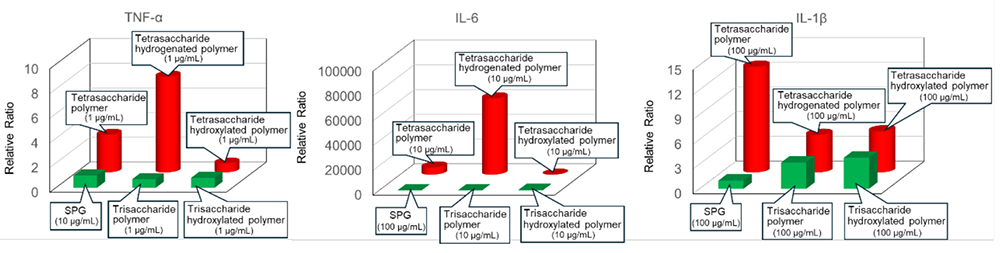
These results indicate that the structural properties of β-glucans—namely, the presence of branched chains and their hydrophobicity—are essential for the activation of immune cells mediated by Dectin-1. These findings highlight the important role of the conformations and physical properties of β-glucan in regulating immune responses.
Future work will focus on optimizing molecular structures to design compounds with even higher immune activation potential, while focusing on controlling induced cytokine profiles. This research can pave the way for the development of more effective immunomodulation molecules.
This study aimed at mimicking the bioactivity of β-glucan, a neutral polysaccharide, by synthesizing β-glucan oligosaccharides, polymerizing glycan units, and modifying the resulting polymers. Dectin-1 is known to be the main immune receptor of β-glucan, but other receptors such as CR3, are also involved, highlighting the need to study these interactions in the future.
The methods adopted in this research contribute to the development of polysaccharide mimic technologies using glycopolymers. The results emphasize the importance of both the sugar structure of the ligand and the polymer backbone in determining immune activation. Although previous designs have selected to use either hydrophilic or hydrophobic backbones in glycopolymer preparation, this study reveals that the effectiveness of such designs depends on the glycopolymer’s interactions with target proteins. Furthermore, the results show that simply aligning ligand sugars is not sufficient for optimal bioactivity.
In the future, polymerization technologies will become increasingly important in the construction of more efficient polysaccharide mimics. It is expected that the continued development of these technologies will allow accurate control of immune responses and support the creation of novel therapeutics.