
Rikiya Taoka
Rikiya Taoka is an Associate Professor of the Department of Urology, Faculty of Medicine, Kagawa University, Japan. He graduated from Kagawa Medical University and obtained his MD in 2001. In 2005, he earned his Ph.D. for his research into inflammation mechanisms from the Graduate School of Medicine at Kagawa University. From 2013 to 2015, he studied as a Visiting Assistant Professor at the University of Texas MD Anderson Cancer Center, concentrating on Cancer stem cell biology. He is currently engaged in research to clarify the anti-cancer effects of Rare Sugars and to introduce them into clinical practice.

Akram Hossain
Akram Hossain, a researcher at Kagawa University’s Faculty of Medicine, earned his MBBS from Mymensingh Medical College, Bangladesh, in 1983 and a Ph.D. from Kagawa Medical University, Japan, in 1995. From 1997 to 2002, he was Assistant Professor (Research) in Gastroenterological Surgery, initiating rare sugar research. He received a postdoctoral fellowship in 2003 from Kagawa Industry Support Foundation to study rare sugars’ physiological functions and safety, and another in 2008 from Industry-Academia-Government at the Cell Physiology Department to continue this work. Since 2018, he has worked in Pharmacology and now Urology, focusing on D-allose’s effects on kidney cancer for clinical use.

Yohei Abe
Yohei Abe is an Assistant professor in the Department of Urology, Faculty of Medicine, Kagawa University, Japan. He graduated from the Kagawa University Faculty of Medicine in 2012. After clinical training in urology, mainly urologic cancer and kidney transplantation, at several hospitals. He entered the Graduate School of Medicine at Kagawa University in 2023 and has been in his current position since April 2024.

Xia Zhang
Xia Zhang is an Assistant Professor in the Department of Urology at the Faculty of Medicine, Kagawa University, Japan. She graduated from Jinzhou Medical University in China, obtaining her MD in 1989. In 2003, she earned her Ph.D. from the Graduate School of Medicine at Kagawa University. From 2004 to 2006, she worked as a specific researcher focusing on rare sugars. Since 2007, she has been serving as an Assistant Professor in the Department of Urology. Her current research focuses on elucidating the anti-cancer effects of rare sugars and translating these findings into clinical applications.

Mikio Sugimoto
Mikio Sugimoto is a Professor of the Department of Urology, Faculty of Medicine, Kagawa University, Japan. He graduated from Kagawa Medical University and obtained his MD in 1988. In 1992, he earned his Ph.D. from the Graduate School of Medicine at Kagawa Medical University. He is a member of many international societies and plays a leading role in prostate cancer. He is currently engaged in research to clarify the anti-cancer effects of Rare Sugars and to introduce them into clinical practice.
Cancer remains one of the leading causes of death worldwide, and its incidence continues to increase. Conventional treatments such as surgery, chemotherapy, radiotherapy, and immunotherapy have advanced significantly; however, their limitations, including toxicity and variable effectiveness, highlight the need for alternative or adjunct therapies. Among emerging approaches, the rare monosaccharide D-allose has garnered attention owing to its potential anticancer properties. This review focuses exclusively on the anticancer effects of D-allose and emphasizes its mechanisms of action, therapeutic benefits, and challenges. As a naturally occurring rare sugar, D-allose has shown promise in modulating cancer cell metabolism, inducing oxidative stress, and influencing immune responses. Preclinical studies have suggested that D-allose exerts anticancer effects by disrupting tumor energy metabolism, promoting oxidative damage, and activating key anticancer signaling pathways. Furthermore, D-allose has been explored for its synergistic potential in enhancing the efficacy of conventional treatments such as chemotherapy and radiotherapy. Novel therapeutic strategies targeting glucose metabolism within the tumor microenvironment have also been discussed, with D-allose emerging as a promising candidate. The review concludes with an outlook on future research directions, advocating for in-depth studies to establish the clinical applicability of D-allose as a part of integrated cancer treatment strategies.
Cancer remains a leading global cause of death, with approximately 10 million fatalities reported in 2020 (World Health Organization), accounting for one in six deaths worldwide1. Common cancers, particularly in the United States and Japan, include colon, lung, prostate, stomach, breast, and liver cancers 2,3. The global cancer burden is expected to increase, reaching an estimated 30 million new cases by 20402,3. Lifestyle factors such as smoking, poor diet, alcohol consumption, and physical inactivity contribute to nearly one-third of all cancer cases4,5.
The increasing prevalence of obesity and type 2 diabetes mellitus further amplifies cancer risk, with chronic conditions such as hyperglycemia, hyperinsulinemia, and inflammation promoting tumor progression6-8. Excessive caloric intake, which often leads to obesity, is closely linked to various cancers, emphasizing the importance of preventing weight gain as a key strategy for reducing cancer risk6,9-11). Sugar plays an important role in this process. The increase in the global consumption of high-glycemic foods has been associated with metabolic disorders and increased cancer risk through mechanisms such as hyperinsulinemia and insulin-like growth factor activation12,13. As a result, efforts to regulate glucose metabolism and reduce the dietary glycemic load are gaining momentum as cancer prevention strategies.
Among these strategies, the use of rare sugars such as D-allose is promising, which is a naturally occurring low-glycemic monosaccharide that has demonstrated unique biological properties in both experimental studies14-16 and clinical trials17. Unlike traditional sugars, D-allose inhibits cancer cell proliferation and metastasis through oxidative stress modulation, apoptosis induction, and angiogenesis suppression12,18-20. Furthermore, studies have suggested that D-allose enhances cancer cell sensitivity to therapies and may serve as a standalone therapeutic agent. Therefore, this review aimed to highlight the anticancer mechanisms of D-allose, focusing on its role in cancer cell metabolism and its potential as a safe and targeted therapeutic intervention.
D-allose, a rare C-3 epimer of D-glucose, is gaining attention owing to its biological and therapeutic potential, which naturally occurs in trace amounts in plants such as Protea rubropilosa and Veronica filiformis21,22. D-allose has the molecular formula C6H12O6. As a low-calorie sweetener, D-allose provides 80% of the sweetness of sucrose; however, only 0.3% of its caloric energy, making it an attractive alternative for dietary and therapeutic applications14. It typically appears as a white, odorless, water-soluble powder with a melting point of 90°C23.
D-allose is classified as a rare sugar by the International Society of Rare Sugars because of its low natural abundance and complex structure24. The Izumoring strategy has enabled efficient enzymatic and microbial production, overcoming natural scarcity and expanding its study25,26. Preclinical and clinical studies have confirmed its safety, with 91.2% excretion in rat urine within 24 h and 67% excretion in humans, indicating minimal metabolic retention or toxicity27.
D-allose exhibits notable biological activities, including anticancer potential, by disrupting glycolysis-dependent energy production in cancer cells28. It also modulates immune responses by inhibiting cytokine production and regulating signaling pathways, making it relevant to inflammation and immune regulation29. Additionally, D-allose has protective effects against ischemia-reperfusion injury30 and therapeutic potential for stroke and hypertension31. Owing to its safety profile, selective targeting of pathological processes, and versatility in metabolic, immune, and cancer therapies, D-allose holds promise as a tool in modern medicine. Ongoing research continues to uncover the underlying mechanisms, positioning it as a candidate for diverse therapeutic applications.
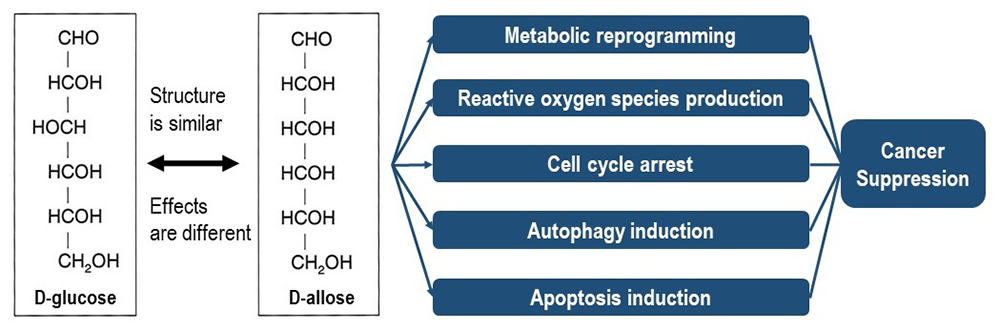
The anticancer potential of D-allose was first reported in 200532 and has shown therapeutic effects in multiple cancer types, including hepatocellular carcinoma32,33, head and neck cancers34-36, ovarian cancer37, cervical cancer38, prostate cancer39, bladder cancer40, small-cell lung cancer41,42, and leukemia43. Its effectiveness (Figure) has been demonstrated in both in vitro and in vivo models44-46, with studies showing tumor growth inhibition, apoptosis induction, and enhanced efficacy of cancer therapies35.
A key mechanism for the anticancer effects of D-allose is the stimulation of reactive oxygen species (ROS) production in cancer cells40. Oxidative stress disrupts redox balance, promotes apoptosis, and suppresses tumor proliferation35. Additionally, D-allose inhibits glycolysis, depriving cancer cells of energy for growth and aligning with the Warburg effect47,48. It modulates pathways such as AMP-activated protein kinase activation and mammalian target of rapamycin (mTOR) inhibition while activating tumor suppressor proteins such as p5341. D-allose is efficacious in combination therapy. It enhances ROS production during radiotherapy and amplifies cytotoxic effects. When combined with chemotherapeutic agents, including docetaxel, it improves treatment outcomes in head and neck malignancies46. Furthermore, D-allose mitigates cisplatin-induced nephrotoxicity45, thereby addressing a major limitation of chemotherapy. Mechanistic studies have revealed that D-allose induces the expression of thioredoxin-interacting protein (TXNIP), a regulator of redox homeostasis that promotes apoptosis. It also suppresses osteoclast differentiation in Ras264 cells49, suggesting its potential role in limiting cancer-related bone metastasis. Its effect on metastatic processes remains under investigation; however, the modulation of oxidative stress, signaling pathways, and inflammation by D-allose highlights its broad therapeutic promise.
Natural scarcity and limited clinical studies pose challenges; however, advancements in enzymatic synthesis have improved accessibility. Future research focusing on metastatic cancers, optimal dosing, and combination therapies is critical for clinical translation. In summary, D-allose targets cancer cells via multiple mechanisms, including modulation of oxidative stress, glycolytic inhibition, and signaling regulation. Its synergistic effects with radiation and chemotherapy highlight its potential as part of integrated cancer treatments. Continued research will pave the way for innovative therapies to improve cancer outcomes.
D-allose exerts anticancer effects by selectively inhibiting glycolysis, which is a primary energy source for many cancer cells. This inhibition leads to energy depletion in cancer cells while sparing normal cells, enhancing their specificity as therapeutic agents47-49. Preclinical studies, including bladder cancer models, have demonstrated that the oral administration of D-allose significantly suppresses tumor growth. This effect is linked to the upregulation of TXNIP and inhibition of glucose transporter-16, thereby competitively reducing glucose uptake50. D-allose reduced cardiomyocyte hypertrophy by inhibiting glycolysis and lowering intracellular glucose levels, mitigating phenylephrine- and isoproterenol-induced cardiac remodeling in cells and mice51.
D-allose uniquely modulates oxidative stress by inducing the production of ROS in cancer cells, leading to mitochondrial dysfunction and apoptosis. Simultaneously, it preserves the mitochondrial integrity of normal cells, thus enhancing their safety profile40,52. TXNIP, upregulated by D-allose, plays a central role in the redox balance, and its activation increases ROS levels in cancer cells, causing oxidative damage and energy depletion52,53. These dual effects underscore the therapeutic specificity of D-allose and its potential as a safer alternative for cancer treatment. Through ROS induction and glycolytic inhibition, D-allose disrupts mitochondrial function in cancer cells, leading to apoptosis and halting tumor progression. This disruption aligns with the metabolic vulnerabilities of cancer cells, further highlighting the role of D-allose as an anticancer agent.
In addition to its anticancer properties, D-allose exhibits antioxidative and anti-inflammatory effects in non-cancer models, such as ischemia-reperfusion injury. It scavenges ROS, inhibits neutrophil-mediated ROS production, and suppresses inflammation54-56. Murata et al. demonstrated superior ROS inhibition in neutrophils compared to other sugars, underscoring its potential to regulate inflammation and improve the tumor microenvironment (TME)18,55.56. These combined actions suggest that D-allose directly affects cancer cells and reshapes the TME to hinder cancer progression. Ongoing research aims to clarify its molecular mechanisms; however, the unique interplay between glycolysis inhibition, oxidative stress modulation, and mitochondrial disruption makes D-allose a promising candidate for metabolism-based cancer therapy.
Radiation therapy, a key cancer treatment, often faces limitations owing to cancer cell resistance. D-allose, a rare sugar, shows promise as a radiosensitizer that amplifies the effects of radiation while providing additional therapeutic benefits. Hoshikawa et al. demonstrated that pretreatment of head and neck cancer cells with D-allose before radiation (2–6 Gy doses) significantly reduced cell proliferation compared with either treatment alone36. This effect was linked to increased expression of TXNIP) a tumor suppressor that disrupts the redox balance enhances oxidative stress and promotes apoptosis57. In vivo studies validated the radiosensitizing potential of D-allose. Combining D-allose with radiation and docetaxel in a preclinical head and neck cancer model significantly suppressed tumor growth, with 50% of the tumors disappearing entirely and the rest showing no regrowth during observation46. The mechanisms underlying the effects of D-allose include its ability to enhance radiation-induced ROS production, amplify deoxyribonucleic acid (DNA) damage, and impair repair processes in cancer cells while sparing normal tissues. D-allose also modulates the TME, boosts inflammatory responses, and increases the efficacy of radiation. Unlike conventional radiosensitizers, D-allose has a favorable safety profile, combining anticancer effects with antioxidant protection in normal tissues. Its ability to enhance both radiation and chemotherapy makes it a versatile option for multimodal cancer therapy.
In conclusion, D-allose represents a novel radiosensitization approach that effectively enhances radiation-induced cancer cell death. Future studies should explore the mechanisms, dosing strategies, and clinical applications in various cancers to unlock their full potential.
Autophagy, a process for degrading and recycling cellular components, is often upregulated in cancer cells to support survival under stress conditions such as hypoxia or glucose deprivation in the TME58-61. Autophagy helps cancer cells evade cell death; however, its regulation, along with apoptosis, is critical for determining cancer cell fate, making these pathways attractive therapeutic targets62. D-allose can modulate autophagy in cancer cells, enhancing therapeutic efficacy when combined with chemotherapy or radiation. D-allose induces autophagy in Lewis lung carcinoma cells, as evidenced by increased levels of the autophagy marker LC3 II and reduced activity of mTOR, a key autophagy regulator, and its downstream protein Beclin142,61. These findings highlight the role of D-allose in regulating autophagic pathways to inhibit tumor progression. Notably, combining D-allose with hydroxychloroquine, an autophagy inhibitor, reduced tumor growth in a dose-dependent manner42, suggesting that D-allose induces a form of autophagy that is detrimental to cancer cells, particularly when used in combination with other therapies. However, tumor cells that survived D-allose treatment exhibited heightened autophagic activity, indicating potential resistance mechanisms and underscoring the need for combination strategies targeting autophagy to enhance efficacy.
The TME plays a crucial role in cancer progression, with immune cells acting as key regulators of tumor suppression or growth. Immune cells rely on metabolic reprogramming during activation and differentiation, making them the focus of cancer research63,64.
D-allose, a rare sugar, has the potential to modulate immune functions. Research by Kenjiro Takao et al. revealed that D-allose selectively inhibits cytokine production in plasmacytoid dendritic cells (pDCs) stimulated with immune activators, such as polyuridylic acid or CpG DNA65. This effect is not linked to reduced glucose metabolism or direct cytotoxicity but to the inhibition of the MAPK signaling pathway, which significantly decreases cytokine production. Additionally, D-allose inhibited the internalization of BCG-anti-BCG IgG complexes in conventional dendritic cells (cDCs), a process critical for antigen presentation66, suggesting that D-allose affects phagocytic pathways, influences immune cell activity, and potentially alters tumor immune surveillance and responses to therapies.
The ability of D-allose to selectively induce autophagy in cancer cells and modulate immune cell functions, particularly in the TME, offers a promising approach to overcome cancer cell survival strategies. By targeting glucose metabolism and autophagic and immune activation pathways, D-allose may significantly improve therapeutic outcomes. Further research is essential to elucidate the underlying mechanisms and optimize its use with other treatments.
D-allose, a rare sugar, shows great potential as an anticancer agent owing to its effects on cellular metabolism and immune modulation. Early studies have suggested that it selectively inhibits cytokine production in pDCs and modulates immune responses, highlighting its potential to enhance immune regulation for cancer therapy. Its ability to target cancer stem cell metabolism and the TME further underscores its therapeutic potential.
However, the anticancer mechanisms of D-allose remain unclear, necessitating further research to clarify its effects on tumor biology and immune functions. Clinical trials are essential to evaluate their safety, efficacy, and applicability in various cancer types. With a deeper understanding of its mechanisms, D-allose could become a valuable addition to cancer therapy, offering innovative strategies to target the metabolic and immune pathways driving tumor progression.
Rikiya Taoka: Conceptualization; Validation; Writing, reviewing, and editing. Akram Hossain; Writing the original draft. Yohei Abe: writing, review, and editing. Zhang Xia: writing, review, and editing. Mikio Sugimoto: Project administration.