
Yuichi Uchino
Vice Director, Keishin Gotanda Eye Clinic
Specially-appointed Lecturer, Department of Ophthalmology, Keio University School of Medicine (Outpatient Clinic for Corneal Disease and Dry Eye)
March 2001: Graduated from the Keio University School of Medicine.
April 2001: Joined the Department of Ophthalmology, Keio University School of Medicine, Assistant Professor.
June 2004: Practicing Assistant Professor, Department of Ophthalmology, Tokyo Dental College Ichikawa General Hospital (Corneal Fellow).
September 2009: Chief, Department of Ophthalmology, Tokyo Electric Power Company Hospital.
April 2012: Harvard Medical School, Massachusetts Eye and Ear, Schepens Eye Research Institute, Postdoctoral Fellow.
April 2016: Full-time Lecturer, Department of Ophthalmology, Keio University School of Medicine (Outpatient Clinic for Corneal Disease and Dry Eye).
April 2022: Current position.
In Japan, dry eye is a very common disease, affecting approximately 20% of the Japanese population and approximately 65% of office workers who use computers for a long time1. While Japan has its own definition (Table 1) and diagnostic criteria2 (Table 2) for dry eye, other Asian countries use the same definition and diagnostic criteria as that of Japan as a result of the exchange of opinions through the Asia Dry Eye Society. It can be said that tear film instability on the ocular surface is of paramount importance in understanding the pathophysiology of dry eye. Tear film instability is involved in the collapse of the glycocalyx on the ocular surface; the roles of galectin 3, a glycocalyx constituent protein, has been increasingly attracting attention. In this article, we briefly review the latest clinical and basic research on tear film behavior, explore the differences in the action of galectin 3 on the ocular surface between the normal and dry eye conditions, and also mention the pathophysiology of the tear film instability.
| Dry eye is a multifactorial disease characterized by unstable tear film causing a variety of symptoms and/or visual impairment, potentially accompanied by ocular surface damage. |
| I) Dry eye subjective symptoms such as eye discomfort and impaired vision | II) A tear film breakup time (BUT) of ≤5 seconds |
The first approach used to detect tear film instability is a fluorescein staining test to visualize the tear film, which is a simple but important method useful in clinical settings. In the normal eye, the tear film is thin measuring approximately 5 to 10 µm in thickness on the ocular surface (Fig. 1). In dry eye, the tear film becomes unstable in several seconds, even if it is stable just after eye opening, and dark areas appear where the tear film is extremely thin (Fig. 2). The time from eye opening to tear film collapse is known as the tear breakup time (BUT). A BUT of ≥10 seconds is judged to be normal, and a BUT of ≤5 seconds is indicative of dry eye. When the BUT is 5 to 10 seconds, special attention should be paid to the fact that environmental factors and even minor changes in the ocular surface are likely to change the BUT to ≤5 seconds.
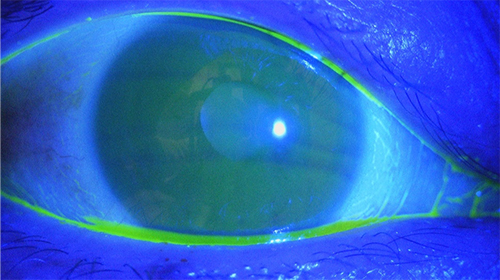
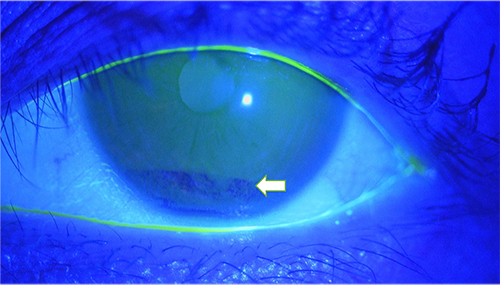
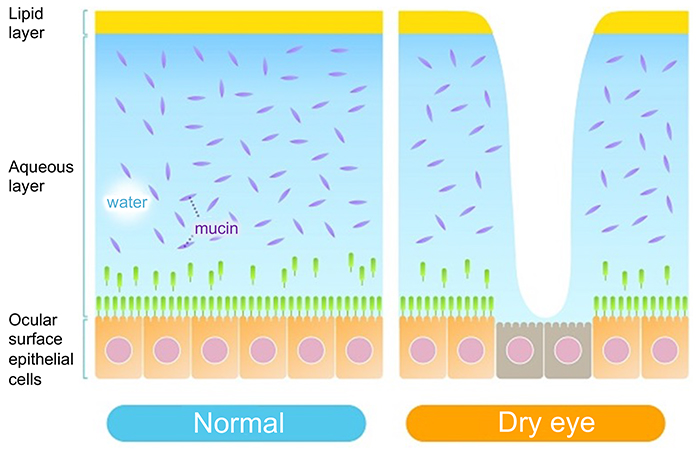
Prior to explaining the mechanism of this rapid collapse of the tear film, the normal ocular surface tear film is described below. The tear film consists of an lipid layer and aqueous layer arranged in this order from the eye surface (Fig. 3). A classical textbook describes the tear film as a three-layer structure whose deepest layer of mucin lies directly on the corneal epithelium surface. However, since the mucin layer was found to comprise two different types of mucin, i.e., secreted mucin in the aqueous layer and membrane mucin expressed on the epithelial side, it is no longer considered part of the layer structure of the tear film. The aqueous layer is particularly important in the tear film. Above all, the largest part of tear film thickness, the aqueous layer, is responsible for transporting all proteins, including electrolytes, antibacterials (lactoferrin, lysozyme, etc.), immunoglobulins (secreted IgA, IgG, etc.), growth factors (EGF, TGF-β, etc.), as well as differentiation inducers (vitamin A, etc.), nutrients, and oxygen essential for keratoconjunctival epithelial cells. In addition, secreted mucins are released from goblet cells in the conjunctiva and occur in solution in the aqueous layer. The secreted mucin, MUC5AC, in particular, is retained in conjunctival goblet cells and released in a timely manner into the tear film, where it retains water in the aqueous layer by forming a crosslinked structure with a large number of mucin-specific glycans. In addition, secreted mucins improve the wettability of the ocular surface by lowering the surface tension of the tear3. This is of paramount importance for tear film stabilization without easy collapse on the ocular surface.
On the other hand, the lipid layer has a number of functions such as stabilization of tear film thickness and suppression of tear water evaporation. Consisting of polar lipids and non-polar lipids (cholesterol esters), the lipid layer is covered by non-polar lipids on its superficial side and contains polar lipids on its lower side to interact with the aqueous layer of water. The non-polar lipids are secreted by the meibomian glands in the eyelid, whereas the polar lipids are mostly secreted by lacrimal glands; as such, the components and structure of the lipid layer remain balanced despite its extreme thinness.
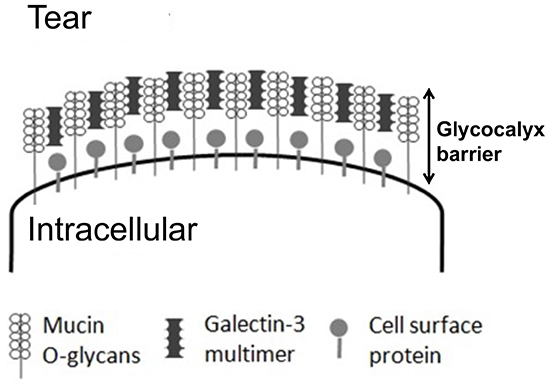
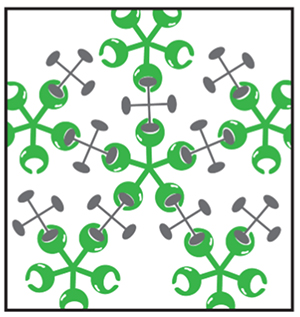
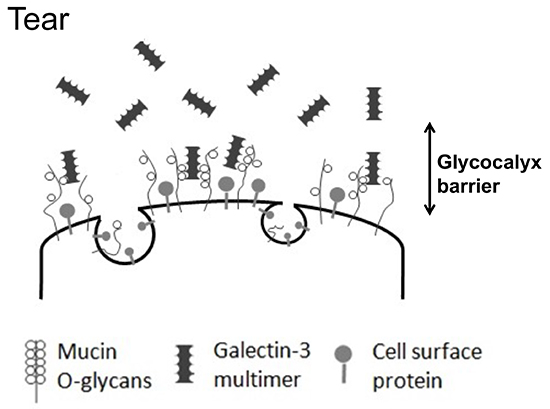
The keratoconjunctival epithelium surface has protruding structures known as microplicae, whose tips include highly expressed membrane mucins that take the form of lawn patterns (Fig. 4). Membrane mucins have a hydrophobic transmembrane domain that is immobilized by the epithelial cell membrane. While the intracellular domain is short, the extracellular domain is much longer, measuring approximately 200 to 500 nm. Corneal epithelial cells have the membrane mucins MUC1, MUC4, and MUC16 expressed therein. MUC16, in particular, is a very long core protein with many glycans, and hence has an extremely large molecular weight. Membrane mucin glycans, like secreted mucin glycans, retain water within their bridging structure and are largely involved in barrier function based on their large size. However, membrane mucins on the ocular surface alone are drifting unsteadily in the tear film like kelp swaying in oceanic currents on a shallow sea bed, and cannot be said to have barrier function. In fact, to join membrane mucins together into a barrier, a protein called galectin 3 binds to membrane mucin glycans, specifically MUC1 and MUC16, which are expressed in large amounts in the microplicae (Fig. 5). Galectin 3 has a polymer-forming site for joining the glycan-binding glycan recognition site and galectin, where membrane mucins and polymerized galectin 3 bind sequentially to form a lattice structure known as the galectin-glycan lattice formation (Fig. 6)5. As described above, membrane mucin is 200 to 500 nm long and has a large number of type-O glycans; therefore, the lattice structure with membrane mucin and galectin 3 forms a barrier of constant thickness known as the glycocalyx on the ocular surface6 (Fig. 7). By serving as a cushion on cell surfaces to protect cells against chemical and physical damage and connecting intercellular spaces, the glycocalyx functions as a highly stable barrier. On the ocular surface, in particular, constant contact with the palpebral conjunctiva produces friction, which is lessened by the glycocalyx. As such, membrane mucins protect our eyeballs at the outside frontier of the eye along with the tear film by retaining water on cell surfaces with large numbers of glycans, acting as a physicochemical barrier of glycocalyx, and reducing friction on the ocular surface.

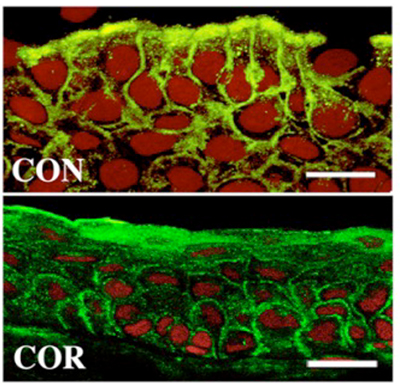
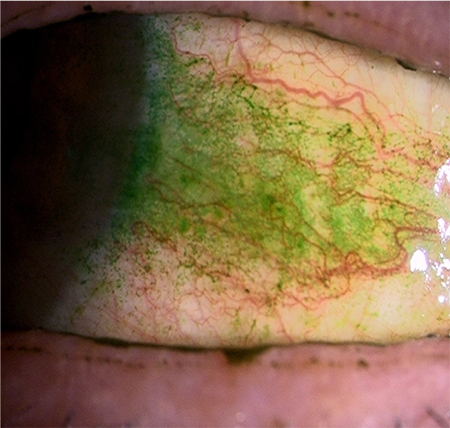
If the glycocalyx barrier collapses, the ocular surface becomes stainable with dyes such as rose bengal and lissamine green (Fig. 8); this method of staining is also used to clinically diagnose dry eye. If the ocular surface glycocalyx barrier is collapsed, galectin 3, which together with membrane mucins should otherwise form the glycocalyx, will also be unstable. Galectin 3 is produced by keratoconjunctival epithelial cells and released into the tear film. On the normal ocular surface, membrane mucin is expressed in large amounts on microplicae and has many glycans to capture galectin 3; therefore, galectin 3 is unlikely to drift freely in the tear film (Fig. 5). However, in dry eye, as the protein expression of MUC16, the predominant membrane mucin on the ocular surface, is lower (Fig. 9), galectin 3 produced by epithelial cells cannot be retained as a portion of the glycocalyx but is released in its free state into the tear film8 (Fig. 10). Hence, it has become evident that tear fluid galectin 3 concentrations are significantly increased due to glycocalyx barrier collapse in diseases, such as dry eye, when compared with the normal condition. On the other hand, galectin 3 production by epithelial cells compared between normal individuals and patients with dry eye was shown to be the same by realtime polymerase chain reaction (PCR) and did not increase significantly in patients with dry eye; but the abundance of galectin 3 in the tear film did increase due to glycocalyx barrier collapse9.
Severity of dry eye can be assessed by scoring the epithelial disorder on the ocular surface. Using tear samples collected from normal individuals and patients with dry eye, we measured and compared tear galectin 3 protein concentrations in three groups classified by severity of dry eye, and found that tear galectin 3 concentrations increased with higher severity. The galectin 3 protein concentration in the tear film was also found to be significantly correlated not only with epithelial disorder score, but also with the amount of tear fluid secretedand subjective symptom severity. It thus proved to be useful in determining the severity of dry eye10.
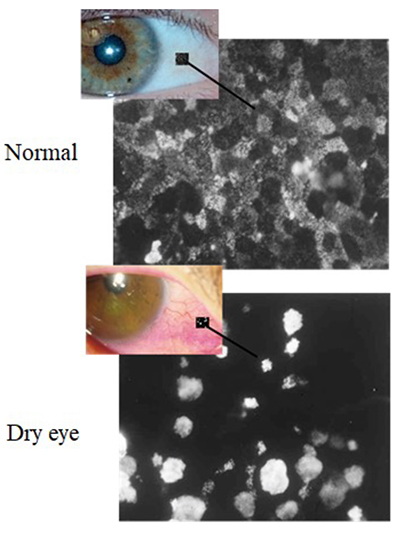
After the galectin 3 content of the glycocalyx was found to be important as described above, many researchers conducted vigorous studies with the expectation of demonstrating the utility of galectin 3 as a dry eye therapeutic. In fact, we hear that a researcher was granted a patent for dry eye treatment and a pharmaceutical company has already started clinical testing in preparation for market launch. When studying in the United States, I performed experiments in the expectation that the glycocalyx barrier function of cultured corneal epithelial cells could be enhanced by administering galectin 3 at various concetrations. Corneal epithelial cells exhibit inflammatory reactions upon addition of IL-1β. I expected that enhancement of the glycocalyx barrier by galectin 3 would lessen the inflammatory reactions following the administration of IL-1β. However, the reverse results were obtained; the intensity of the inflammatory reactions induced by IL-1β increased as the concentration of external galectin 3 increased11. Galectin 3 might coagulate cell surface receptors to prolong receptor-mediated reactions; however, our experiments did not demonstrate it. However, I found that tagged galectin 3, when administered to cultured corneal epithelial cells, was absorbed in the cells. Although absorption might have some impact on the promotion of inflammatory reactions, details remain unknown.
In my presentation about the above research at an academic meeting held outside Japan, I received a comment from a respected scientist who was the owner of patents on galectin 3 as a dry eye therapeutic. “Your presentation today was wonderful. But one of my patents is gone. That’s bad.” I remember her smiling face at that time. Worsened inflammation due to galectin 3 may actually be purposeful on the ocular surface. A high level of galectin-3 in tear fluid is likely to damage epithelial cells and markedly reduce the expression of membranous mucin. New multi-layers of epithelial cells cannot cover the ocular surface unless the cells are metabolically active; taking into account cell losses and migrations, intercellular adhesion molecules such as tight junction molecules are unnecessary at least temporarily. Inflammation provides critical molecules for increasing the mobility of the epithelium, including intercellular adhesion molecules. Transient promotion of inflammation is no longer necessary after epithelial cells restore the eye to its normal state. This promotive action may be another key role of galectin 3 in glycocalyx collapse.
The membrane mucin MUC16 is as important in the construction of the glycocalyx barrier as galectin 3. Actually, a drug that maintains a high level of this MUC16 protein expression in corneal epithelial cells is commercially available as an ophthalmic suspension for dry eye. Its nonproprietary name is rebamipide. Since its market launch as an oral medicine for gastric ulcers in 1990, it has been widely prescribed as a therapeutic mainly for mucosal protection. The ocular surface is also a mucosal membrane, and dry eye is often accompanied by epithelial disorders. To respond to this situation, rebamipide was developed as an ophthalmic suspension in 2012. Comparing the effect of different concentrations of rebamipide on the protein expression levels of the membrane mucins MUC1, MUC4, and MUC16 in multi-layered cultured corneal epithelial cells, we found that rebamipide raised MUC16 protein expression levels12. Clinical research following its market launch found that rebamipide ophthalmic suspension markedly ameliorated subjective symptoms in patients with dry eye, i.e., pain and sensation of foreign body, not easily mitigated by conventional dry eye ophthalmic suspensions13. The alleviation of symptoms such as pain and sensation of foreign body is probably attributable to decreased friction between the eye and eye lid as a result of MUC16 protein-induced re-construction of the glycocalyx barrier.
The glycocalyx on the ocular surface consists of a lattice structure of the membrane mucin MUC16 and multimerized galectin 3. In patients with dry eye, the MUC16 protein expression decreased, the barrier was broken, and galectin 3, which should otherwise form the glycocalyx, was released into the tear fluid at increased levels. In addition, in cases of severe dry eye with intense barrier collapse, the tear galectin 3 concentration increased significantly. It was confirmed that recovery from glycocalyx collapse and alleviation of subjective symptoms such as pain and sensation of foreign body were due to the use of rebamipide ophthalmic suspension, which acts via normalization of the expression of the membrane mucin MUC16.