
Eiji Miyoshi
Eiji Miyoshi MD and PhD, Professor and Chairman, Department of Molecular Biochemistry and Clinical Investigation, Osaka University Graduate School of Medicine
History: I graduated from Osaka University Graduate School of Medicine in 1986. After clinical training of Gastroenterology and Hepatology, I have been studying glycobiology. I became a professor in 2007 and was a dean of Osaka University Allied of Health Science in 2014-2015. I have been a director of the Center for Borderless Design of Medicine in Osaka University.
Research Interests: Research directed at understanding the biological functions of sugar chains at the cellular and molecular level and clinical investigation to many human disease including cancer, inflammation, DM and liver disease.
The liver is the largest organ in the human body. It weighs approximately 1,500 grams and consists of 250 billion cells, including hepatocytes (70-80%) and nonparenchymal cells (20-30%). The biological function of hepatocytes is production of serum proteins, lipid/glucose metabolism, detoxification, and production of bile. Nonparenchymal cells include Kupffer cells (hepatic macrophage-like cells), hepatic stellate cells (vitamin A-storage cells), bile duct epithelial cells and endothelial cells. The liver is called the “silent” organ because patients manifest no symptoms even when 70% of hepatic function is disrupted. However, patients with acute hepatitis show severe general fatigue and jaundice. Inflammation and regeneration of the liver in chronic hepatitis leads to liver cirrhosis, the end stage of chronic liver disease. In decompensated liver cirrhosis, patients show icterus, ascites and hepatic encephalopathy. Characteristic of chronic liver diseases is the step-by-step progression of chronic hepatitis and liver cirrhosis to hepatocellular carcinoma (HCC). While hepatocellular carcinoma is rarely detected in healthy individuals, 5-8% patients with liver cirrhosis develop HCC. The cause of chronic hepatitis is infection by type B or type C hepatitis virus. Hepatitis B virus (HBV) is a DNA virus and infection can be prevented by vaccination while hepatitis C virus is an RNA virus, infection by which cannot be prevented by vaccination. Recently chronic hepatitis C has been completely cured with DAA (direct acting antiviral) treatment. Compared with conventional interferon treatment, treatment with DAA is much more effective with low side effects. However, DAA is expensive. In contrast, interferon and nucleotide analog are used for the treatment of type B hepatitis. Since the effectiveness of interferon is low and patients must take a nucleotide analog for life, a novel therapy for type B hepatitis is required. Other types of hepatitis besides viral hepatitis include alcoholic liver disease, drug-induced hepatitis and autoimmune hepatitis. In Japan, as a result of changes in life style to resemble more those of Western nations, the number of patients with non-alcoholic fatty liver diseases (NAFLD) has risen. 10-20% of NAFLD patients suffer non-alcoholic steatohepatitis (NASH), which develops into liver cirrhosis followed by HCC. Pathological diagnosis by liver biopsy is the only established method that can differentiate NASH from NAFLD. However, there are 10-20 million NAFLD patients in Japan and it is impossible to perform liver biopsies on all NAFLD patients. A noninvasive biomarker for NASH diagnosis is required.
Recent advances in methodology of glycomics have given rise to a variety of glycol-biomarkers. Comprehensive oligosaccharide analysis of sera showed alteration of hepatic glycoproteins and/or immunoglobulin. A search for key words “glycosylation” and “biomarker” in PubMed found approximately 4000 articles, a third of which discussed glycol-biomarkers for liver diseases. It is well known that the oligosaccharide structure is dramatically changed by cancer and inflammation. The glycosylation patterns of hepatic glycoproteins change as a result of liver inflammation and cancer/cancer metastasis. One of the most representative glycol-biomarkers for HCC is fucosylated AFP (AFP-L3) 1, 2. AFP-L3 was developed in Japan and the first HCC biomarker recognized by the USFDA (United States Food and Drug Administration) 3. While alpha-fetoprotein (AFP) is a conventional cancer biomarker for HCC, AFP levels are also elevated in patients with chronic hepatitis and liver cirrhosis. In contrast, AFP-L3 level is specific to HCC patients. Dr. Shinji Satomura has written an interesting Internet review on the subject.
In the 1990s, the present author and his group succeeded in the purification and cDNA cloning of several glycosyltransferases involved in the synthesis of N-glycan branching (Fig. 1). Surprisingly, expression of these glycosyltransferases is quite low in normal liver but increases in chronic liver disease and HCC 4, 5. In general, it is thought that fucosylation, sialylation and branching are three major changes in glycosylation associated with carcinogenesis. As shown in Fig. 1, N-acetylglucosaminyl transferase V (GnT-V) is involved in the synthesis of b1-6 GlcNAc branching leading to tri- and tetra N-glycan formation. N-glycan branching formation results in increases in terminal fucosylation and sialylation. GnT-V is one of the most important glycosyltransferases involved in cancer metastasis 6. One reason why expression of GnT-III, GnT-V and Fut8 is quite low may be that most hepatocytes in normal liver are at the G0 stage of the cell cycle. When cells proliferate, oligosaccharide modification by these glycosyltransferases plays a pivotal role in growth factor receptor signaling. Percial hepatectomy model of rats induces up-regulation of GnT-III, GnT-V and Fut8 7. There are many reports on glycosylation and receptor signaling. A review of liver regeneration and glycosylation by Dr. Tomohiko Fukuda scheduled for publication in April 2019 summarizes the content and detailed molecular mechanisms.
In 2003-2007, the NEDO MG project (leader: Professor Hisashi Narimatsu of AIST) developed a novel glycan biomarker, M2BPGi (mac-2 binding protein glycosylation isomer), which is associated with liver fibrosis 8. Glycomic technology using lectin microarray was used to develop glycol-biomarkers for several diseases in the NEDO MG project. M2BPGi is a WFA lectin-binding Mac-2 binding protein (Mac-2bp) and is in use clinically as a biomarker for liver fibrosis covered by Japanese health insurance. The mission of the NEDO MG project was to find a cancer biomarker using cancer cell lines and sera of cancer patients. However, the marker for liver fibrosis was the first to be found. Long experience in liver and glycosylation research has shown that the most dynamic changes in expression of glycosyltransferases are observed at the stage of liver cirrhosis 9. Thus it is natural that M2BPGi was the first glycol-biomarker to be found in the NEDO MG project. Mac-2bp is a serum protein with elevated levels in patients with cancer and viral infection. Mac-2bp is a ligand of galectin 3 in serum. After M2BPGi was developed as a biomarker for liver fibrosis, many reports on the subject have been published. M2BPGi might be used for biomarkers for conditions besides liver fibrosis. The detailed procedure for the development of M2BPGi and its clinical application are described in a review by Drs.Yoshihiro Kamada and Eiji Miyoshi on this same Internet site. Comprehensive analyses of serum oligosaccharides showed dramatic changes in glycosylation patterns 10. In HCC, patients have chronic hepatitis and liver cirrhosis. Therefore, glycosylation patterns of HCC patients can be compared to those of chronic hepatitis or liver cirrhosis patients to recognize characteristic glycosylation of HCC. Carrier proteins of oligosaccharides characteristic of HCC should be identified. Since IgG is a major serum glycoprotein, changes in IgG glycosylation are observed in many inflammatory diseases. It also changes with aging. Galactose-deficient IgG (agalactosyl IgG) was first found in patients with rheumatoid arthritis including autoimmune diseases 11. Agalactosyl IgG can be used as a novel biomarker in inflammatory bowel diseases to follow clinical disease activity 12, while IgG glycosylation determination of lectin binding can be a biomarker for liver fibrosis 13.
A genetic approach seeking an HCC biomarker detected glypican 3. Glypican 3 was originally found to be a gene involved in intestine differentiation with differential display by Filmus et al. 14. Glypican 3 is a proteoglycan having long heparansulfate chains, and mutation of glypican3 was found in a human congenital disorder 15. Expression of glypican3 is significantly higher in HCC tissue than in liver fibrosis, and glypican 3 is secreted after shedding. Serum glypican 3 levels determined by ELISA can be a novel biomarker for HCC 16. Interestingly, Nakatsura et al. have developed a novel vaccine using glypican 3 peptide. This is described elsewhere on this Internet site.
The above has shown that the oligosaccharide structure of the liver changes dramatically with disease progression, leading to marked changes in serum glycosylation pattern. Until recently, many researchers appear to have avoided conducting glycobiology research because of the technical difficulties involved. However, advances in glycan analytical methods have drawn clinical researchers to become glyocobiologists. Many glycan studies have been reported at clinical conferences, and there exist a number of large grants for the study of glycobiology. Please see the reviews of Drs. Kouji Ogawa and Naoya Sakamoto planned for publication in June 2019 on this Internet site. At the annual meeting of the Japanese Society of Carbohydrate Research in 2018, “Studies in carbohydrate research present and future” was discussed. One famous researcher in glycobiology noted that the discipline of glycobiology could disappear within 20 years because anyone would be able to easily conduct glycobiology experiments. If this comes true, hepatologists may eventually have too many research sources in glycobiology to handle.
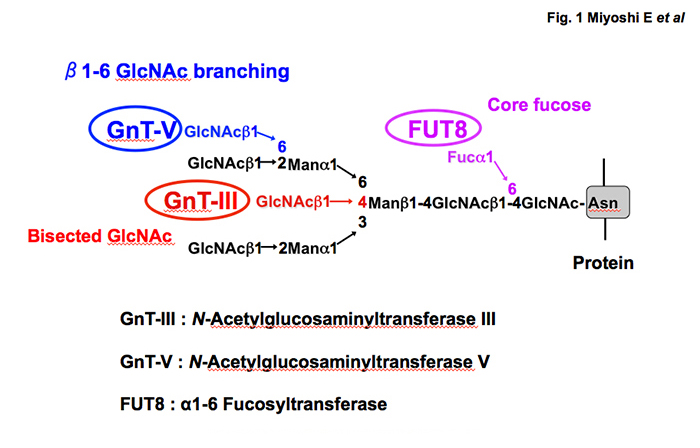
Fig. 1 Three major glycosyltransferases involved in N-glycan branching and their products
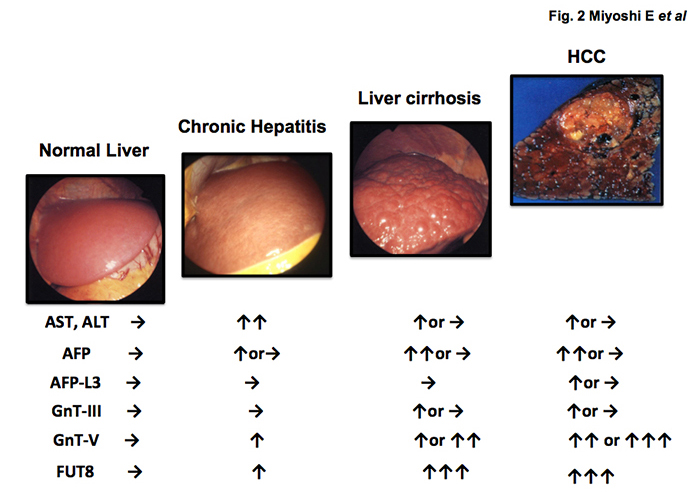
Fig. 2 Changes in liver enzymes (AST/ALT), AFP, AFP-L3 and glycosyltransferases involved in N-glycan branching (GnT-III, GnT-V, FUT8) in hepatocarcinogenesis