
Koichi Honke
Dr. Honke graduated from Hokkaido University School of Medicine obtaining his M. D. in 1983. Since he started his research in the laboratory of Professor Akira Makita, Cancer Institute, Hokkaido University Medical School in 1984, he has been studying the metabolism and function of glycolipids. He obtained his Ph. D. in 1994 from Hokkaido University. He worked at Osaka Medical Center for Maternal and Child Health as a chief researcher from 1995 to 1999. He was then promoted to associate professor of the Department of Biochemistry, Osaka University Medical School in 1999. His major contribution has been the purification, molecular cloning and gene targeting of the sulfotransferase responsible for the biosynthesis of sulfoglycolipids and the discovery of the β Gal 3-O-sulfotransferase gene family.
Life maintains harmony by continuous cross-talk between divergent elements everywhere within an organism. These cross-talk occurs at every rank of the hierarchy of life, such as inter-molecules, inter-cells, inter-tissues, inter-organs and inter-organisms, and elements at each rank, forming a complex network. According to the theory of Complex Dynamical Systems, certain macroscopic phenomena emerge from interactions between microscopic elements via nonlinear, large-scale interactions. This is called self-organization. A higher rank phenomena are assumed to be generated based on the principle.
The hierarchy of life contains many ranks where organisms are formed from genomes that are the design of life. When it comes to glycogenes, which are responsible for the biosynthesis of carbohydrate chains, there are seven ranks: glycogenes, glycan-synthesizing enzymes, glycans (glycoconjugates), molecular complexes (cell formation), intercellular networks (tissue formation), intertissual networks (organ formation), and interorganic networks (organism formation) (Fig. 1). We are not able to fully understand the biological processes if any of these ranks is skipped. Therefore, it is not easy to elucidate the entire mechanism underlying biological events, even if some abnormal phenotype is observed at the levels of tissue formation to organism formation (the fifth to seventh rank) resulting from disruption of a gene. Although it takes much time and work, only steady studies that accumulate findings at each rank and link these individual events with related changes at the neighboring rank will enable us to learn the truth.
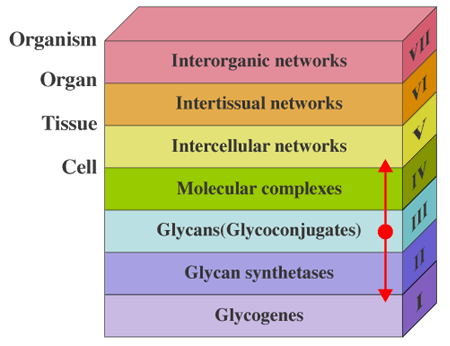
Fig. 1 The hierarchy of life.
Glycans are elements belonging to the third rank of the hierarchy (Fig. 1). If we start to explore at the rank of glycans, we can easily go up and down to other ranks. From this point of view, glycans may be the most suitable subject to study self-organization in life. The author believes that the theme of this series “Beyond Glycogenes” aims to comprehend the biological functions of carbohydrate chains from this viewpoint.
The author has been studying biological functions of sulfoglycolipids. Two major sulfoglycolipids exist in mammal: one being the sulfatide, which is a sphingolipid and the other being the seminolipid, which is a glycerolipid1, 2 (Fig. 2). Sulfatide is a major lipid component of the myelin sheath and is synthesized in myelin-generating cells1. Seminolipid is synthesized in spermatocytes and maintained in the subsequent germ cell stages2. The fact that the distribution of sulfoglycolipids is tissue-specific suggests that their biological roles play out at the sites where they are expressed1, 2.
The carbohydrate moiety of sulfatides and seminolipids has the same structure and is biosynthesized via sequential reactions catalyzed by common enzymes: ceramide galactosyltransferase3 (CGT, EC 2.4.1.45) and cerebroside sulfotransferase4, 5 (CST, EC 2.8.2.11) (Fig. 2). CGT is located in the endoplasmic reticulum and CST is found in the Golgi membranes. It has been proved that neither enzyme has isozyme as evidenced by gene disruption in mice6-8. The tissue distribution of sulfoglycolipids is mainly regulated by the tissue-specific expression of the CGT gene. CST gene expression is also tissue-specific, but its regulation is not as strict as the CGT gene9. Studies on the expression mechanism of sulfoglycolipids in oligodendrocytes and spermatocytes, and on the gene expression mechanisms of the CGT and CST genes mean going down the ranks of the hierarchy shown in Fig. 1. The association of the regulatory mechanisms of gene expression of the CGT and CST genes in these tissues is an intriguing question from the viewpoint of the molecular evolution of sulfoglycolipids.
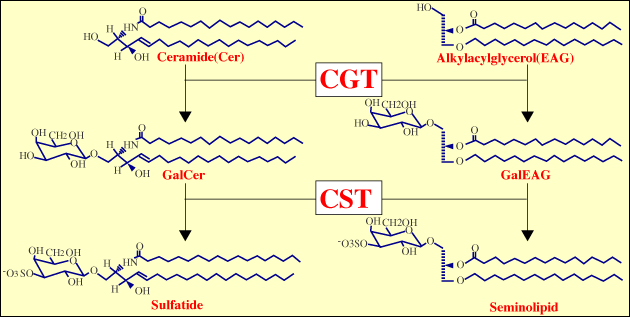
Fig. 2 Structures and biosynthetic pathways of sulfatides and seminolipids
Mammals have acquired the property known as myelin during the evolution so as to enhance the conductivity of the neuronal impulse. Oligodendrocytes produce vast amounts of myelin, a unique, lipid-rich biomembrane with a relatively simple array of myelin-specific proteins in the central nervous system. This membrane, an extension of the oligodendrocytes plasma membrane, forms multilamellar, spirally wrapped sheaths around neuronal axons. The gaps between adjacent myelin sheaths are referred to as nodes of Ranvier, and myelin forms lateral loops there10 (Fig. 3). These myelin loops terminate at the paranode region and engage in the formation of a septate-like adhesive junction with the axon membrane, axolemma. This specialized axo-glial junction acts as an electronical and biochemical barrier between nodal and internodal membrane compartments. Voltage-gated sodium channels concentrate in the nodal axolemma, while shaker-type K+ channels, Kv1.1 and Kv1.2, localize within the juxtaparanodal axolemma. Saltatory conduction of the action potential is attributed to this organization. The adhesion of myelin to the axolemma plays a critical role in this clustering of ion channels. Thus myelin serves not only as a simple insulator but also as a functional platform of the neuron-glia interaction. Myelin is generated in Schwann cells in the peripheral nervous system.
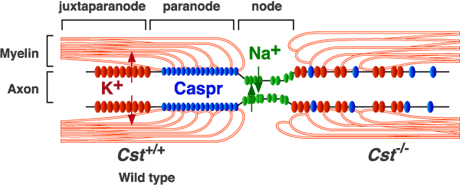
Fig. 3 Organization of nodes of Ranvier and their alteration in CST-deficient mice.
In wild-type mice (left side), myelin membranes and axon membranes form junctions at the paranode region and Na+ and K+ channels cluster at the node and juxtaparnode regions, respectively, separated by the septate-like axo-glial junction. In CST-deficient mice (right side), formation of the axo-glial junction is disordered and the clustering of Na+ and K+ channels is also disturbed.
GalCer and sulfatide comprise 23% and 4% of the total lipid content in the myelin sheath, respectively1. These glycolipids are sequentially synthesized by the action of CGT on the precursor, ceramide in the endoplasmic reticulum, and subsequently by the catalysis of CST in the Golgi apparatus in oligodendrocytes and Schwann cells (Fig. 2). GalCer and sulfatide are first expressed at a critical point during oligodendrocyte differentiation, when progenitors cease to proliferate and commence terminal differentiation. This leads to the prediction that these glycolipids play a significant role in the regulation of oligodendrocyte differentiation and myelin function.
In order to elucidate the physiological function of sulfoglycolipids, CST-deficient mice were created by means of gene targeting8. CST-deficient mice show a complete loss of sulfatide in brain but no changes in other glycolipids, including GalCer. CST-null mice were born healthy, but began to display hindlimb weakness by 6 weeks of age and subsequently showed a pronounced tremor and progressive ataxia. Histological analysis revealed that axons were well myelinated in CST-null mice, but myelin vacuolation and focal axonal swelling (axonal spheroid formation) were observed, as also noted in CGT-null mice6, suggesting secondary neuronal degeneration due to abnormalities in myelination. Electron microscopic analysis of myelinated nerve fibers revealed disorganized termination of the lateral loops at the nodes of Ranvier, similar to that reported in CGT-deficient mice11 (Fig. 3). Furthermore, clustering of Na+ and K+ channels at the nodes is also deteriorated in CST-null mice12 as observed in CGT-null mice13 (Fig. 3). These findings strongly suggest that sulfatide is an indispensable molecule for the adhesive junction of myelin loop and axolemma at the paranode region. It has been reported that three proteins: Caspr/paranodin, contactin, which are on the axolemma, and the 155 kDa splice isoform of neurofascin (NF155), which is on the myelin membrane, form the axo-glial adhesion apparatus at the paranodal region10. How a lipid component of myelin, sulfatide, interacts with these protein complexes and maintains the adhesive junction is an important problem for future studies.
Despite significant neurological disorders, CST-null mice are able to survive to more than one year of age8. The phenotype of the CST-deficient mice was milder than that of CGT-deficient mice6, 7 in terms of the age of onset, life span, and the severity of symptoms. Moreover, CST-deficient mice show neither decrease in the maximal conduction velocity or demyelination, unlike CGT-null mice8, 11. This discrepancy suggests that sub-localization on myelin sheath is different between GalCer and sulfatide and that GalCer acts not only as a precursor for sulfatide synthesis, but also as a functional molecule.
Since sulfatide emerges when oligodendrocyte progenitors cease dividing and begin differentiating, it is assumed to be involved in the regulation of oligodendrocyte differentiation. In fact, oligodendrocyte differentiation is enhanced in CGT-KO mice14. Furthermore, oligodendrocyte differentiation in wild-type mice was found to be blocked by anti-sulfatide antibody but not by anti-GalCer antibody12. These findings strongly suggest that sulfatide is a key negative regulator of the oligodendrocyte differentiation. This hypothesis was confirmed by the fact that terminal differentiation of oligodendrocytes is enhanced in CST-KO mice (unpublished observation by Hirahara et al.). Hence sulfatide plays a role in the regulation of oligodendrocyte differentiation, in addition to their eventual roles as structural components of mature myelin.
Spermatogenesis occurs in the seminiferous tubules in the testis15 (Fig. 4). Spermatogonia that are germline stem cells sit in a special place on the basement membrane of seminiferous tubules called the niche. These spermatogonial stem cells differentiate into spermatocytes, ceasing proliferation. Subsequently, spermatocytes undergo meiosis twice and differentiate into haploid cells referred to as spermatids, migrating from the surrounding area to the lumen of the seminiferous tubules on the stroma cells termed Sertoli cells with mutual interaction. After that, they mature into spermatozoa via morphogenesis and leave the seminiferous tubules for the epididymis through the efferent ductules.
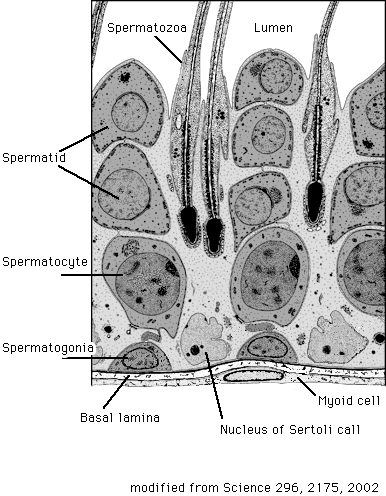
Fig. 4 Spermatogenesis in a seminiferous tubule.
( modified from Science 296, 2175, 2002 )
More than 90% of glycolipids in the testis consists of a unique glyceroglycolipid, seminolipid2. Its carbohydrate moiety is the same as that of sulfatide and the synthesizing enzymes are found to be common6-8 (Fig. 2). Seminolipid is highly synthesized at an early stage of spermatocytosis.
In CGT-deficient mice, neither galactosylalkylacylglycerol (GalEAG) nor seminolipid is synthesized, and spermatogenesis is arrested prior to meiosis16. On the other hand, primary spermatocytes seem to be normal in CST-deficient mice, but spermatogenesis is blocked at the metaphase of the first meiosis8. The arrested stage in the germ cell differentiation of CST-null mice appears to occur somewhat later than in CGT-null mice, suggesting that both GalEAG and seminolipid are successively involved in the genetic program of spermatogenesis in the same order as their biosynthesis. Alternatively, GalEAG that lacks a sulfate group may compensate for the seminolipid to a certain extent. Since these glycolipids are expressed on the cell surface of primary spermatocytes from the end of the leptotene stage or the zygotene stage and later, the interaction between Sertoli cells and spermatocytes, which is known to be important for their differentiation, may be disrupted in these knockout mice.
Our studies gradually revealed that sulfoglycolipids are essential for organisms. Now the chance has come to commence research on self-organization involving sulfoglycolipids. The first target will be the rank where sulfoglycolipids are one of the elements, and the first approach will be the search for molecules interacting with sulfoglycolipids. To identify molecules interacting with certain carbohydrate structures at a given site in brain and testis is not as easy as previously thought, because genuine oligosaccharide receptors exist in only limited quantities and in confined regions. In the case of sulfoglycolipids, they are known to bind specifically to diverse molecules such as laminin, selectins and anti-coagulant factors in vitro1, but it is meaningless unless the molecules that interact with sulfoglycolipids are identified at the sites where sulfoglycolipids are proved to function, namely in axo-glial junction and spermatocyte-Sertoli cell communication. To this end, conventional procedures to purify active materials from whole homogenates of organs could not be employed, because there are many molecules that specifically bind sulfoglycolipids but are irrelevant to the function. Therefore, we must develop a novel method to identify the genuine molecule using a limited amount of materials. If we could identify such a molecule, the next step would be to reconstitute a molecular complex containing sulfoglycolipids. In addition, we must examine whether the deletion mutant of the target molecules shows a similar phenotype to the mutant-lacking sulfoglycolipids. After clearing all these hurdles, the higher rank may come into sight.