
Naoyuki Taniguchi
Professor Taniguchi graduated from the Hokkaido University School of Medicine and obtained M.D. in 1967, and completed the doctoral course of medicine at the Graduate School of Hokkaido University and obtained Ph.D in 1972. He was then appointed an assistant professor at the Hokkaido University School of Medicine in 1975 and appointed a visiting associate professor, Department of Biochemistry Cornell University Medical College, New York at the laboratory of Dr. Alton Meister between 1976 and 1977. He was appointed an associate professor, Graduate School of Environmental Science, Hokkaido University in 1977 and later appointed an associate professor, Biochemistry Laboratory Cancer Institute, Hokkaido University School of Medicine in 1980 at the laboratory of Dr. Akira Makita. He was appointed as a Professor and Chairman, Department of Biochemistry, Osaka University Medical School in 1986. He received a Grant in Aid for Scientific Research on Priority Areas entitled "Sugar remodeling and cellular communications" from the Ministry of Education, Science Sports and Culture, Japan between 1998 and 2000. He is an honorary member for the American Society for Biochemistry and Molecular Biology. He will receive International Glycoconjugate Organization (IGO) Award, and Docteur Honoris Causa of the University Henri Poincaré in 2001. The president for the Annual Meeting of the 74th Japanese Biochemical Society in 2001. Editorial board members: J. Biol. Chem., Glycobiology, Glycoconjugate J., Biochem. Biophy. Res. Commun. (from March 2002).
At the present time approximately 110 glycogenes which encode glycosyltransferases and related genes have been cloned. Even though some of the biological functions of those genes have been elucidated, most of the actual functions of these genes continue to be obscure, and limited information is available in terms of their pathophysiolgical significance. Therefore in order to clarify the functional significance of these genes, one of the major strategies is focused on the identification of likely target molecules in vivo and the identification of their functional significance.
Knock out or transgenic mice have already been reported for several glycogenes and currently available information indicate that some are lethal and some lead to interesting phenotypic changes (see review in refs 1-2). Moreover different types of patients with CDG (Congenital Disorders of Glycosylation) have been reported (3-4). These facts clearly indicate that the glycosylation of proteins, glycolipids and proteoglycans play major roles in development, differentiation and oncogenesis. However questions are raised as to the nature of the actual target molecules in vivo. Phenotypic changes observed in these studies or symptoms of patients with CDG sometimes fail to explain the likely target molecule(s) and, therefore, to explain the pathophysiology of the KO or transgenic mice or CDG. The reason why it does not explain the pathophysiology is that even if a glycogene is knocked out or is lacking or overexpressed, phenotypic change(s) are not due to the direct effect of the knocked out or deleted glycogene or the overexpressed glycogene, but, rather, due to the indirect effect of target molecule(s) in which glycosylation has been diminished or enhanced.
The functional glycomics comes of age in the postgenomic era.
The post-genomic research era has now been launched and science has become more concerned with the proteomic approach, including the post-translational modification of proteins. However, most proteome projects have not adequately discussed the analytical and functional details of the glycosylation of proteins, lipids and proteoglycans. Therefore in order to understand real biology, the introduction of concept of a more integrated form of glycobiology and glycotechnology, called "glycomics" seems appropriate. This field of research will focus on the analytical details of glycosylation as well as the functional details of glycosylation. According to the recent data based on Swiss-Prot data bases described by Apweiler et al (5), more than half of all proteins undergo glycosylation in eukaryotic cells and this would be also true in the case in prokaryotic cells.
Several groups have already initiated work on the glycomic approach or glycome studies (see in Proteomics 1, 2001, Post-translational modifications. Guest editor :Nicolle H Packer and ref 6-14) and this, in fact, represents one of the major projects in the post-genomic research era. Paulson et al (15) have recently organized a consortium for functional glycomics which will focus on the carbohydrate binding proteins.
One of the likely strategies in the area of functional glycomics is to identify the protein by 2DGa and in-gel digestion with glycanases such as PNGase Fb to release glycans, followed by in-gel digestion with a protease such as trypsin. For protein analysis, extracted peptides can be subjected to MALDI-TOF MSc. For the analysis of glycan sugars or oligomers are labeled by attaching a fluorescent molecule, and subjected to HPLC analysis using exoglycosidase digestion as proposed by Rud et al (9). Moreover, in order to understand the function, gene transfection of the gene of interest in vitro would be also useful strategy for functional glycomics, as has been reported by our group (6). This approach may permit an understanding of the “glycome” of cells as it relates to the function of the proteins. It is also important to emphasize the use of a specific lectin staining or lectin columns or a specific antibody against carbohydrate moiety to identify the glycosylated proteins. These strategies would be applicable for “Functional glycomics” of N-glycans, O-glycans, GPI anchors and proteoglycans.(Fig. 1,Fig. 2)
We sincerely hope that functional glycomics will open a new venue for the postgenomic research era in life sciences.
a peptide-N(4)-(N-acetyl-β-D-glucosaminyl) asparagine amidase F
b matrix assisted laser desorption/ionization-time of flight-mass spectrometer
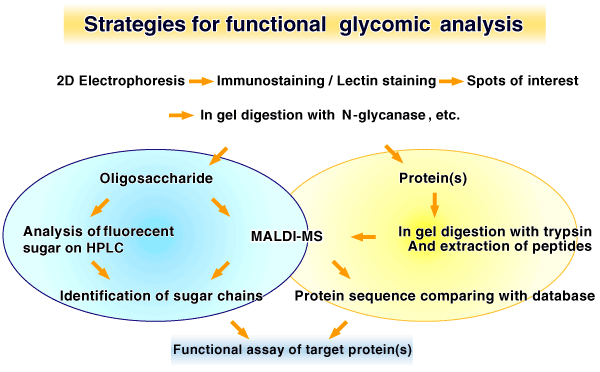
Figure 1
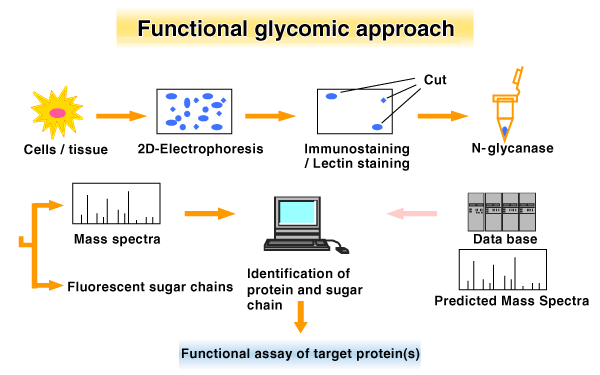
Figure 2