 |
Calnexin (CNX) and calreticulin (CRT) are homologous lectin-like molecular chaperones that interact with newly synthesized glycoproteins through specific monoglucosylated oligosaccharides to regulate quality control of glycoproteins in the endoplasmic reticulum (ER) (1). In spite of similar sugar-binding specificity, CNX and CRT bind to a variety of distinct target proteins. Gene knockout studies demonstrated that CRT-knockout mice were lethal, and CNX-knockout mice showed early death with severe neural abnormalities, indicating that CNX and CRT could not compensate each function in development. These also suggest that CNX and CRT play distinct biological roles in the cell.
The structure of calnexin/calreticulin
CNX is an 88-90 kDa transmembrane protein of type I in the ER. CRT, another homologous ER resident, is a 55-60 kDa soluble protein with the C-terminal amino acid sequence -K-D-E-L, a retrieval signal in the ER. Both are monomeric, calcium-binding proteins, and are related members of the legume lectin family. Based on structural and functional studies, CNX/CRT can be divided into three distinct domains; N-terminal [N], proline-rich [P], and C-terminal [C] (Fig. 1). The N-domain, a globular 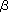 -sandwich domain with homology to legume lectin, has been modeled based on crystallographic data for CNX. The P-domain shows a characteristic structure with an extended and curved arm connected to a globular N-domain. The domain contains two characteristic repeats 1 (-I-x-D-P-(D/E)-(A/D)-x-K-P-(E/D)-D-W-D-(D/E)-) and 2 (-G-x-W-x-x-P-x-I-x-N-P-x-Y-), and the sets are repeated four times in CNX, and three times in CRT. The N-terminal region encompassing the N and P-domain of CNX/CRT interacts with misfolded proteins and glycoproteins, binds ATP, Zn2+ and Ca2+ with high affinity and low capacity, and is likely to be involved in the chaperone function of the proteins. The P-domain is also important for binding to ERp57 that is a thiol-disulfide oxidoreductase to assist the disulfide exchange reactions in misfolded proteins in the ER. The C-domain of CRT binds Ca2+ with high capacity and plays a role in Ca2+ storage in the ER in vivo. CNX has a transmembrane sequence and a following cytoplasmic domain that can undergo phosphorylation by casein kinase II to interact with ribosomes. -sandwich domain with homology to legume lectin, has been modeled based on crystallographic data for CNX. The P-domain shows a characteristic structure with an extended and curved arm connected to a globular N-domain. The domain contains two characteristic repeats 1 (-I-x-D-P-(D/E)-(A/D)-x-K-P-(E/D)-D-W-D-(D/E)-) and 2 (-G-x-W-x-x-P-x-I-x-N-P-x-Y-), and the sets are repeated four times in CNX, and three times in CRT. The N-terminal region encompassing the N and P-domain of CNX/CRT interacts with misfolded proteins and glycoproteins, binds ATP, Zn2+ and Ca2+ with high affinity and low capacity, and is likely to be involved in the chaperone function of the proteins. The P-domain is also important for binding to ERp57 that is a thiol-disulfide oxidoreductase to assist the disulfide exchange reactions in misfolded proteins in the ER. The C-domain of CRT binds Ca2+ with high capacity and plays a role in Ca2+ storage in the ER in vivo. CNX has a transmembrane sequence and a following cytoplasmic domain that can undergo phosphorylation by casein kinase II to interact with ribosomes.
The calnexin/calreticulin cycle in glycoprotein folding
In the biosynthesis of glycoproteins bearing N-glycans, the oligosaccharide Glc3Man9GlcNAc2 is attached to the asparagine (N) residue contained in the consensus sequence N-x-S/T of newly synthesized polypeptides in the ER. The oligosaccharides are processed by glucosidase-I and II, after which CNX/CRT binds to the Glc1Man5-9GlcNAc in glycoproteins (Fig. 1). The sugar-binding (lectin) site that interacts with the stretch of Glc1Man3 is located on the concave surface of the 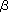 -sheet sandwich part in the globular N-domain of CNX/CRT. If the glycoprotein is completely folded in the ER, the terminal glucose is removed by glucosidase-II and the glycoprotein is released from the CNX/CRT chaperone cycle. However, if the glycoprotein is not properly folded, the terminal glucose is once again attached by the action of UDP-glucose: glycoprotein glucosyltransferase (UGGT), which discriminates between folded and unfolded substrates (2). Then re-glucosylated substrates again get back to the CNX/CRT chaperone cycle. Together, CNX and CRT form a specific chaperone cycle, the “CNX/CRT cycle”, for the biosynthesis of glycoproteins in the ER (Fig. 2). Although the lectin activity is no longer questioned in the functions of CNX/CRT, the significance of protein-protein interactions between CNX/CRT and substrates is still under controversy (1). If proper folding is not completed, misfolded glycoproteins with high-mannose type oligosaccharides lacking glucose will be retro-translocated out of the ER to cytosol via the interaction with EDEM/Htm1/Mnl1 and/or Derlin-1, then degraded by proteasomes (i.e. ER-associated degradation [ERAD], see GlycoWord QS-A02). -sheet sandwich part in the globular N-domain of CNX/CRT. If the glycoprotein is completely folded in the ER, the terminal glucose is removed by glucosidase-II and the glycoprotein is released from the CNX/CRT chaperone cycle. However, if the glycoprotein is not properly folded, the terminal glucose is once again attached by the action of UDP-glucose: glycoprotein glucosyltransferase (UGGT), which discriminates between folded and unfolded substrates (2). Then re-glucosylated substrates again get back to the CNX/CRT chaperone cycle. Together, CNX and CRT form a specific chaperone cycle, the “CNX/CRT cycle”, for the biosynthesis of glycoproteins in the ER (Fig. 2). Although the lectin activity is no longer questioned in the functions of CNX/CRT, the significance of protein-protein interactions between CNX/CRT and substrates is still under controversy (1). If proper folding is not completed, misfolded glycoproteins with high-mannose type oligosaccharides lacking glucose will be retro-translocated out of the ER to cytosol via the interaction with EDEM/Htm1/Mnl1 and/or Derlin-1, then degraded by proteasomes (i.e. ER-associated degradation [ERAD], see GlycoWord QS-A02).
Other cellular functions of CNX and CRT
CNX and CRT are mainly localized in the ER for the quality control of glycoproteins but are sometimes detected on cell surface. CRT is also detected in extracellular space and cytosol. In addition to the participation in glycoprotein folding, CRT is involved in many biological processes including the regulation of Ca2+ homeostasis, intracellular signaling, cell adhesion, gene expression, and nuclear transport (3). Outside the ER, the biological relevance of lectin functions of CNX or CRT remains an open question.
|
|
|
| References |
(1) |
Helenius A, Aebi M: Roles of N-linked glycans in the endoplasmic reticulum. Annu. Rev. Biochem., 73, 1019-1049, 2004 |
|
(2) |
Parodi AJ: Protein glucosylation and its role in protein folding. Annu. Rev. Biochem., 69, 69-93, 2000 |
|
(3) |
Michalak M, Corbett EF, Mesaeli N, Nakamura K, Opas M: Calreticulin: one protein, one gene, many functions. Biochem. J., 344, 281-292, 1999 |
|
|
|
Links
|
|
GP-B07 Sweet ER Quality Control: The Role of N-glycosylation in Protein Quality Control (Sato Sachiko) |
|
|
GP-C04 N-linked Oligosaccharide as a Signal for Proteolysis (Tadashi Tai) |
|
|
ES-C01 Distribution and Function of Lectins in Organisms (Jun Hirabayashi) |
|
|
 -Mannosidase-like Protein Involved in Quality Control in the Endoplasmic Reticulum(ER) (Nobuko Hosokawa, Kazuhiro Nagata) -Mannosidase-like Protein Involved in Quality Control in the Endoplasmic Reticulum(ER) (Nobuko Hosokawa, Kazuhiro Nagata) |
|
|
Cytoplasmic Peptide:N-Glycanase (Tadashi Suzuki) |
|
|
|
|
|



