

 |
 |
Regulation of Immune Responses by Galectins |
|||||||||||||||||||||||||||||
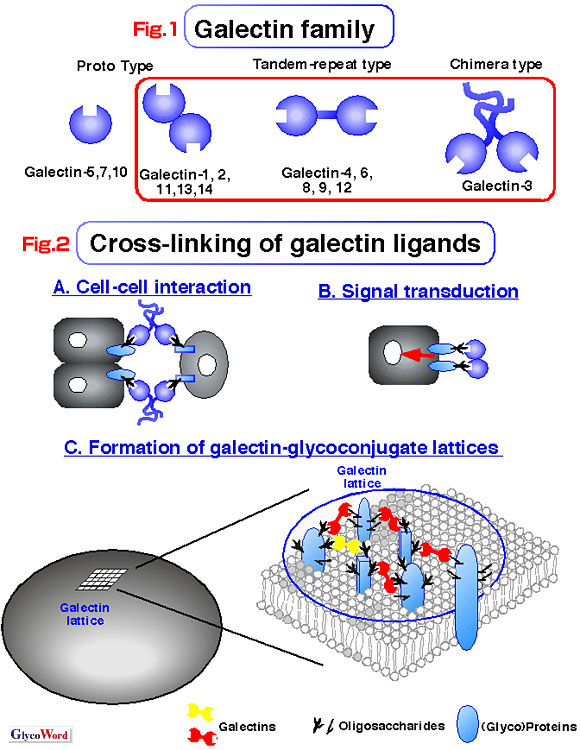
| The immune response system is composed of both innate and acquired immunity, both of which participate effectively in a cooperative manner to protect body integrity. It has been well established that various lectin families, such as the selectin and collectin families, play critical roles in the immune response. Almost all of the mammalian lectins that are involved in immunity are membrane proteins. Interesting exceptions are galectins, the topic of this section, and collectins, which can distinguish various pathogens from host cells. Galectins are an ancient family of proteins defined by their affinity for 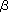 -galactoside and by conserved sequence elements. Many galectins have been identified in vertebrates as well as in some invertebrates (Fig.1). Although they are not sorted through the classical secretory pathway, it is known that galectins are secreted/released from cells when immune responses are triggered. Once released, galectins can autocrinely or paracrinely modulate immune responses by cross-linking galectin ligands on various immune cells. Various immunomodulative activities of galectins have been established, suggesting that galectins act as proinflammatory cytokines, and some of these activities will be introduced here (note: galectins are also implicated in various intracellular metabolism, which will not be discussed here). As demonstrated for various cytokines, galectins' immunomodulative activities also display functional pleiotropy and redundancy. In the case of cytokines, the pleiotropy/redundancy partly resides in the fact that cytokines often share common signal transduction molecules to transmit signals. As for galectins, the pleiotropy/redundancy appears to reside in galectins' mutivalency in ligand binding. Because of this multivalency, soluble galectins could cross-link the surface ligands in at least three different ways that are not mutually exclusive, leading to the mediation of cell-cell interactions, agonistic signal transduction and the formation of galectin lattices (Fig.2). Specifically, unlike other cytokines, it should be noted that galectins can act as adhesion molecules for leukocytes, or alternatively can form galectin-glycoconjugate (glycoprotein/glycolipid) lattices on the surface of leukocytes. The immunological importance of galectin lattices have recently attracted attention, since the galectin lattices could robustly restrict the lateral mobility of surface receptors, raising the threshold for ligand-dependent receptor clustering and signal transduction. -galactoside and by conserved sequence elements. Many galectins have been identified in vertebrates as well as in some invertebrates (Fig.1). Although they are not sorted through the classical secretory pathway, it is known that galectins are secreted/released from cells when immune responses are triggered. Once released, galectins can autocrinely or paracrinely modulate immune responses by cross-linking galectin ligands on various immune cells. Various immunomodulative activities of galectins have been established, suggesting that galectins act as proinflammatory cytokines, and some of these activities will be introduced here (note: galectins are also implicated in various intracellular metabolism, which will not be discussed here). As demonstrated for various cytokines, galectins' immunomodulative activities also display functional pleiotropy and redundancy. In the case of cytokines, the pleiotropy/redundancy partly resides in the fact that cytokines often share common signal transduction molecules to transmit signals. As for galectins, the pleiotropy/redundancy appears to reside in galectins' mutivalency in ligand binding. Because of this multivalency, soluble galectins could cross-link the surface ligands in at least three different ways that are not mutually exclusive, leading to the mediation of cell-cell interactions, agonistic signal transduction and the formation of galectin lattices (Fig.2). Specifically, unlike other cytokines, it should be noted that galectins can act as adhesion molecules for leukocytes, or alternatively can form galectin-glycoconjugate (glycoprotein/glycolipid) lattices on the surface of leukocytes. The immunological importance of galectin lattices have recently attracted attention, since the galectin lattices could robustly restrict the lateral mobility of surface receptors, raising the threshold for ligand-dependent receptor clustering and signal transduction. |
|||||||||||||||||||||||||||||
 |
|||||||||||||||||||||||||||||
| During immune responses or inflammation, leukocytes are first mobilized to the affected regions. The leukocytes phagocytose invading foreign cells or dead cells, and secrete various factors including microbicidal factors and cytokines, which initiate other immune cascades. During the responses, apoptosis is induced in some damaged leukocytes or in subsets of activated T cell populations to maintain self-integrity. During and after the responses, the affected sites require functional restoration either through resolution or repair. Thus, angiogenesis and wound healing processes are initiated at the affected sites. Various recent reports have established that through cross-linking of galectins' ligands, galectins could influence all of those responses. Leukocytes, such as neutrophils, macrophages, and lymphocytes, are recruited from blood stream to the affected sites. Different kinds of chemoattractants facilitate efficient vectorial migration of leukocytes to the sites. Galectin-3 is a chemoattractant for monocytes and macrophages but not for neutrophils, while galectin-9 uniquely attracts eosinophils. To achieve the efficient migration of leukocytes, coordinated regulation of multiple sets of adhesion molecules is required. Some galectins (ex. galectin-1, 3, 8, and 9) are implicated in the cell migration process. Interestingly, galectins could induce biphasic effects (adhesion-promoting and anti-adhesion) depending on the conditions. Adhesion-promoting effects could result from direct cross-linking of ligands expressed on different cells (A) or from the activation of various adhesion molecules such as integrins though galectin-mediating signal transduction (B). In contrast, some of anti-adhesion activity may result from leukocyte surface galectin lattices. Galectin-1 and 3 trigger oxidative burst in neutrophils, leading to release of reactive oxygen intermediates that potentially kill invading pathogens, and galectin-3 induces degranulation of mast cells. Cytokine production is also modulated or triggered by galectins. For example, by inhibiting Th1 cytokines such as TNF- Some galectins are implicated in leukocyte homeostasis through induction of cell death by apoptosis. For example, extracellular galectin-1 induces apoptosis in subsets of T cell populations (such as immature thymocytes or activated T cells), and B cells as well as macrophages, while galectin-9 induces apoptosis of eosinophils and some T cell lineages. Possibly because of galectin-1's apoptotic nature, the introduction of galectin-1 in various autoimmune model systems results in the amelioration of clinical symptoms. During or after infection/inflammation, the wound healing process and angiogenesis are promoted partly to restore the affected regions. Recent works have demonstrated that galectin-3 and -7 accelerate re-epithelialization in the wound healing process. Galectin-3 could stimulate capillary tube formation, which could induce angiogenesis. When galectins are released and paracrinely bind to the neighboring cells, galectins exhibit various roles to modulate immunity. Inside cells, galectins are segregated from their glycoconjugate ligands, which are present in the secretory pathway and on the plasma membrane. When severe infection or chronic infection/inflammation occurs, galectins could either be released from cells damaged by inflammation/infection or actively secreted from differentiated stromal cells, alveolar resident macrophages in the lung or inflammatory macrophages emigrated in the setting of chronic infection/inflammation. Thus, galectins are released from cells when the human defense system should receive several "danger signals" to initiate immunity. It is intriguing to propose that galectins act as molecules that can send "danger signals" to immunity and that should thus, one day, be categorized as a new cytokine group— the lectinocytokine. |
|||||||||||||||||||||||||||||
| Sachiko Sato (Glycobiology Laboratory, Research Centre for Infectious Diseases, Laval University Medical Centre, Québec, Canada) | |||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
| Jan. 15 , 2004 | |||||||||||||||||||||||||||||
|
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||