|
Fluorescence Polarization-Carbohydrate-Protein Interaction in Solution
|
|
|
 |
Fluorescence polarization (FP) is a technique for analyzing the interaction of carbohydrate-protein, antigen-antibody or protein-protein in solution. FP is easy and simple because it does not require immobilization or washing step, and it is used in immunoassay, and therapeutic drug monitoring in clinical pharmacy. The theory of FP was established by Perrin in 1926 (1).
Fluorescent molecules in solution, excited with a plane-polarized light, emit light into a fixed plane if the molecules remain stationary during the fluorophore's excitation. However, the molecule will emit light into a different plane if the molecule rotates and tumbles during the fluorophore's excitation. Therefore, when a fluorescent molecule binds to a large molecule such as protein, the emitted light is obviously less depolarized (Fig.1).
|
|
 |
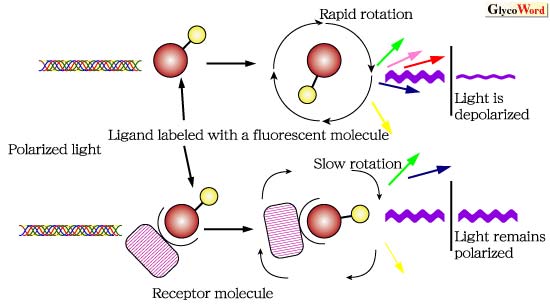 |
Fig. 1 Principle of Fluorescence Polarization.
|
|
 |
|
|
|
Polarization value means the extent of molecular rotation during the period between excitation and emission. Oligosaccharides, which have a small molecular mass, rapidly move and rotate in solution, and therefore have low polarization values. In contrast, when an oligosaccharide binds to a protein molecule such as lectin, the polarization value has a large value because of the decrease in molecular rotation. Thus, the molecular interaction between carbohydrate and lectin can be evaluated by observing the change in polarization value. Recently, Oda et al. examined carbohydrate-lectin binding using fluorescence polarization technique, and reported that the lectin from saffron bulbs specifically recognizes the N-glycan core structure (2-3)(Fig.2).
|
|
|
|
Fig.2
Specific recognition of the core glycan by the lectin from saffron bulbs. |
|
|
|
Fluorescence polarization technique is a powerful tool for the analysis of carbohydrate-protein interaction due to its easy operation. |
|
|
|
|
|
Mitsuhiro Kinoshita, Yasuo Oda
(Faculty of Pharmaceutical Sciences, Kinki University) |
|
|
|
|
|
| References |
(1) |
Perrin, F. J. Phys. Rad. 1, 390-401, 1926 |
|
(2) |
Oda, Y., Kinoshita, M., Nakayama, K. and Kakehi, K. Biol. Pharm. Bull. 21, 1215-1217, 1998 |
|
(3) |
Oda, Y., Nakayama, K., Abdul-Rahman, B., Kinoshita, M., Hashimoto, O., Kawasaki, N., Hayakawa, T., Kakehi, K., Tomiya, N. and Lee, Y.C. J. Biol. Chem. 275, 26772-26779, 2000 |
|
|
|
|
|
| Mar. 15, 2001 |
|
|
|
|
|
|
|



